Separation and Purification Your Way: siRNA to mRNA
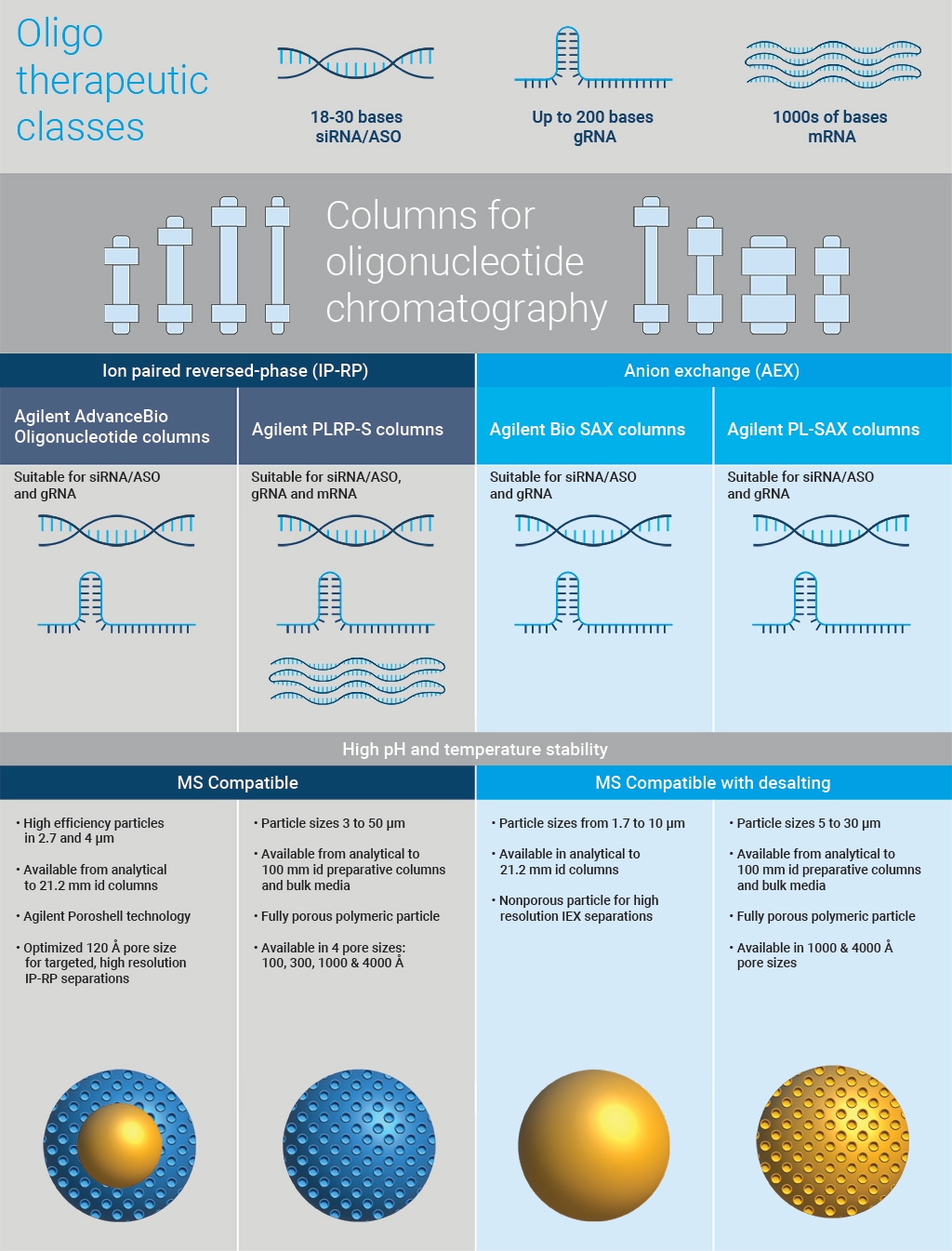
Agilent recognizes the diversity of the oligonucleotide landscape and offers the optimal chemistry for robust resolution, reproducibility, and long column lifetime.
Nucleic Acid Characterization
Agilent AdvanceBio Oligonucleotide columns feature high-efficiency, 2.7 µm and 4 μm superficially porous Poroshell particles. Using technology exclusive to Agilent, we chemically modify the particles to make them exceedingly resistant to high-pH mobile phases. This provides better separation and denaturing conditions for optimal resolution and oligonucleotide separations. We also bond them with an end-capped C18 phase that delivers excellent selectivity for oligonucleotides, making it ideal for LC/UV and LC/MS characterization workflows. In addition, we test every batch of AdvanceBio Oligonucleotide media with a resolution standard to ensure consistent performance.
LC/UV and LC/MS Analysis
Small Oligonucleotide Characterization
- Evaluation of Different Ion-Pairing Reagents for LC/UV and LC/MS Analysis of Oligonucleotides
- Direct Analysis of In-Process Oligonucleotides Without Manual Purification
- Fast and High-Resolution Reversed-Phase Separation of Synthetic Oligonucleotides
mRNA Characterization
Ion-Pair Reversed Phase Purification
Agilent offers two different particle chemistries to support ion-pair reversed phase (IP-RP) analysis of oligonucleotides.
The AdvanceBio Oligonucleotide column family offers a high-efficiency, high-resolution superficially porous particle chemistry that is available in 2.7 µm and 4 μm particle sizes with 100 Å pore size. It is a fully scalable column chemistry platform with dimensional offerings from analytical through 10 mm id semi-prep and 21.2 mm id prep. For high-resolution separations of short synthetic oligos the AdvanceBio Oligonucleotide column chemistry is an ideal choice.
The Agilent PLRP-S polymeric media is physically and chemically robust and therefore well suited to the harsh mobile phase conditions required for IP-RP oligonucleotide purification. Available in a variety of different particles (3 μm to 30 μm) and pore sizes (100 Å all the way up to 4000 Å) with a dimensional offering from analytical through 100 mm id prep, PLRP-S is ideal for longer oligo sequences like mRNA or for larger process-scale preparative purifications.
Together, these two Agilent column portfolios can uniquely support all your oligonucleotide IP-RP separation workflow needs, from early-stage discovery through process scale purifications.
Applications
Agilent offers a range of technical notes and applications to support your method development.
Webinars
Attend our on-demand webinar series discussing BioPharma Purification and Analysis Solutions.
Agilent Solutions
Shop the range of Agilent ion-pair reversed phase columns for oligonucleotide purification.
Easily Scale Up to Purification Using Agilent Oligonucleotide Columns
Watch our short video to learn more
Over the last several decades, there has been continued interest and focus on new therapeutic modalities for the regulation of protein expression at the RNA and even DNA level. The purification of oligonucleotides (ONs) can be particularly challenging with the high similarity of API and impurities.
Agilent recognizes the unique challenges for each oligonucleotide API, offering a range of chemistries, pore size, and dimensions to meet your needs.
Anion Exchange Purification
Featuring 1000 Å and 4000 Å totally porous particles, Agilent PL-SAX columns are uniquely suited for the purification of therapeutic oligonucleotides, utilizing common buffers like Tris-HCl with organic modifiers for gRNA separations, or strong basic conditions for separating oxidation impurities of thiolated oligonucleotides.
Unlike traditional silica and small-pore chemistries, PL-SAX has the pH and temperature stability—paired with optimal pore size—required to separate various oligonucleotide sizes and modifications.

Applications
Agilent offers a range of technical notes and applications to support your method development.
Webinars
Attend our on-demand webinar series discussing BioPharma Purification and Analysis Solutions.
Agilent Solutions
Shop the range of Agilent anion exchange columns and media for oligonucleotide purification.
Meet the Altura family of HPLC columns

Agilent Altura HPLC columns with Ultra Inert technology feature an advanced coating that blocks active metal sites, ensuring an inert flow path while maintaining the strength, pressure tolerance, and consistency of a traditional stainless steel HPLC column.
The result? Altura columns unlock the true separation potential of the stationary phase. Experience superior chromatographic performance, faster equilibration, reduced carryover, and enhanced sensitivity for your most challenging metal-sensitive analytes.
Explore nowIntroducing AdvanceBio Spin columns and 96-sample plates for desalting and buffer exchange
With Agilent AdvanceBio Spin columns and plates, users can easily and quickly remove excess salts or undesired small molecules such as synthesis byproducts from their sample matrix, or exchange the sample matrix between different buffer conditions.
These gel-filtration-based spin columns are available in analytical scale and semipreparative scale columns for single samples, as well as 96-sample plates for high-throughput settings. The column bed contains the same crosslinked dextran particles in all three formats, so users can easily scale up or down in sample number or volume.
Learn moreComplete Workflows to Advance Your Lead Oligonucleotide Candidates from Discovery to Market
Agilent can help with complete workflows featuring instruments, software, columns, and consumables optimized for oligonucleotide analysis. In addition, Agilent can help you with GMP manufacturing of late-stage and commercial-quantity oligonucleotides.
RA44694.6135300926
Agilent offers a range of particle and pore sizes with our PLRP-S polymeric media. From bench to process, PLRP-S scales with your needs.
Scale-up
Purification of Single-Stranded RNA Oligonucleotides Using High-Performance Liquid Chromatography
Column size
Dynamic Binding Capacity of Oligonucleotides on PLRP-S Columns and Stationary Phases
Method Optimization
This application note describes common method parameters that can be optimized when purifying oligonucleotides ranging from 25 to 100 bases in length.
Modified Oligos Separations
High Resolution Separations of Oligonucleotides using PL-SAX Strong Anion-Exchange HPLC Columns
AEX column comparison
This application note compares Bio SAX and PL-SAX. Bio SAX is optimal for high resolution purifications, while PL-SAX is ideal for high binding capacity.
Synthetic oligonucleotides have evolved into a diverse set of molecules that vary greatly in size, structure, and modifications. With this diversity, comes many synthesis related impurities with high homology to the parent molecule.
This series of free training modules discuss the theory of oligonucleotide purification, guiding through chemistry selection, method optimization, scale-up, and API characterization in quick video modules.
Introduction: Designing Your Method
Module 1 discusses the series of variables that should be considered when optimizing a purification method including yield, resolution & throughput.
Impurity Profiles
Synthetic oligonucleotides can range in size from a few bases to 1000's of bases in length. The oligonucleotides can have many impurities associated with this sythesis including truncation, incomplete thiolation, base loss, and adducts. We explore the many impurities that will require purification for a pure API.
Oligonucleotide Size and Pore Selection
With the range of oligonucleotide sizes used as therapeutics, selection of the optimal pore size to give max binding capacity while having large enough pores for effective mass transfer is critical. This module explores performing dynamic binding capacity experiments to determine optimal pore size for your oligonucleotide purification and future scale-up.
Anion Exchange Method Optimization
Anion exchange (AEX) is often selected as a preferred purification method due to its low cost buffer systems that can be readily available for scale-up to manufacturing. This module discusses the optimization of AEX for improved separation by adjusting common variables such as temperature, organic, and pH.
Ion-Pair Reversed Phase Optimization
Ion-pair reversed phase separation is often used for oligonucleotide characterization and purification. Providing high resolution separation, ion-pair reversed phase is often utilized to separate challenging, co-eluting impurities. This module discusses method optimization including temperature, pore size, ion pair selection, and future scale-up.
LC/MS Analysis of Synthetic Oligonucleotides
During oligonucleotide sythesis, purification and final formulation, monitoring of the API for final purity is critical throughput processing. This module explores the use of LC/MS for oligonucleotide characterization, including selecting an optimal ion pair and hexafluoroisopropanol (HFIP) concentration.