Access Agilent eNewsletter April 2016
Fast arsenic speciation for food and urine analysis with Agilent LC-ICP-QQQ
Brian Jackson
Research Associate Professor of Earth Sciences,
Dartmouth College
Amir Liba
Agilent SPSD Application Chemist Team Manager
Inorganic arsenic (iAs) is a class one carcinogen, topping the 2015 Agency for Toxic Substances and Disease Registry (ATSDR) Substance Priority List of pollutants, and a major health hazard worldwide.
The major exposure routes to humans are through water and food consumption, and the major elimination route is through urine. Because arsenic (As) exists in different forms (species) in the environment, speciation analysis is necessary to quantify these individual species. The organic forms of As, principally arsenobetaine (AB), arsenocholine (AC), monomethylarsonic acid (MMA) and dimethylarsinic acid (DMA), are less toxic than iAs (As(III) and As(V)), so regulations mainly focus on the threshold limits for iAs, rather than total As.
High numbers of samples and evolving regulations
Many population based human exposure studies require the measurement of thousands of urine samples, making a fast and reliable routine method essential. Impending EU food regulations for iAs in rice specify limits of 0.2 mg/kg for white rice and 0.1 mg/kg for rice products intended for infants and young children. These new regulations increase the need for high sample throughput iAs analysis, and we forecast that similar regulations may well be forthcoming in the US.
Requirement for faster chromatographic run times
A common analytical method used for As speciation is liquid/ion chromatography (LC/IC) with an anion exchange column, interfaced with collision/reaction cell ICP-MS. Typical run times are 10-20 minutes, depending on the chromatographic method and the range of As species to be separated. For example, the current Centers for Disease Control and Prevention (CDC) method involves a 17-minute runtime [1]. Reducing the chromatographic run time is highly desirable to improve sample throughput.
Short column length and high flow rates yield fast chromatography
Based on a method developed in a previous study [2], an Agilent 1260 Infinity Quaternary LC was fitted with a smaller column—a 50 x 4.1 mm PRP-X100 with a 5 µm pore size—and interfaced to an Agilent 8800 ICP-QQQ. The ICP-QQQ was setup to measure As in MS/MS mode using O2 as the reaction cell gas. The first quadrupole, Q1, was set to 75 amu, to prevent all ions from entering the cell, apart from As and any on-mass (m/z 75) interferences, while the second quadrupole, Q2, was set at 91 amu, allowing for the detection of the reaction product ion AsO+; As reacts quantitatively with O2, while common interferences or doubly charged species do not.
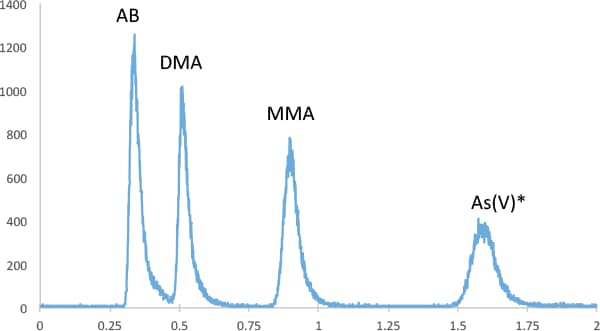
Figure 1. Isocratic separation of four arsenic species at 500 ng/L (ppt). 50 mM (NH4)2CO3 at 1.5 mL/min flow rate. Column temp. of 35 °C. As(V)* is the sum of As(V) and As(III) after oxidation.
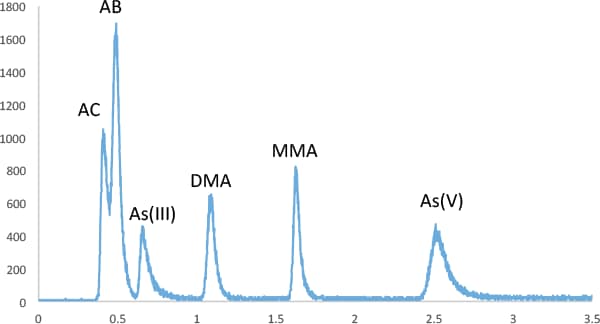
Figure 2. Separation of six arsenic species spiked at 400 ng/L (ppt) in urine, using gradient conditions given in Table 1. Column temp. of 35 °C.
Food samples: complete separation in under 2 minutes
In our test, As(III) was oxidized to As(V) prior to the chromatographic separation. This approach is useful for food analysis, where the dilute acid extraction could promote method-induced changes in the iAs oxidation-state. Also, from a regulatory standpoint, it is only necessary to quantify total iAs (i.e. the sum of As(III) + As(V)) separate from the other species.
We used an isocratic separation with 50 mM (NH4)2CO3 (Eluent A) and a flow rate of 1.5 mL/min to separate AB, DMA, MMA and As(V)*, where As(V)* is the total iAs following oxidation (Figure 1). The separation was complete in 1.7 minutes, and instrument detection limits of ca. 10 ng/L (ppt) for AsB, DMA, MMA, and 50 ng/L for iAs, measured as As(V), were achieved. For quality control of the food analysis we used NIST SRM 1568b rice flour, certified for MMA, DMA and iAs. Recoveries for 1568b (n=3) were 104%, 106%, 96% for DMA, MMA and total iAs, respectively, and the CV of the triplicates was < 10% for each of the species.
Urine samples: complete separation in 3.5 minutes
For As speciation in urine, we used a gradient separation (Table 1) varying both flow rate and eluent strength to separate AC, AB, As(III), DMA, MMA, and As(V) (Figure 2). AC was only partially separated from AB, but these compounds elute in the void volume and are often grouped together as ‘un-retained organic arsenic.’ In practice, we have not detected AC in any of the urine samples analyzed in our laboratory.
| Time, min | Eluent A: 50 mM (NH4)2CO3 in 3% methanol, % | Eluent B: 3% methanol, % | Flow rate, mL/min |
|---|---|---|---|
| 0 | 20 | 80 | 1 |
| 1.45 | 20 | 80 | 1 |
| 0.46 | 100 | 0 | 2 |
| 2.5 | 100 | 0 | 2 |
| 2.51 | 20 | 80 | 1 |
| 3.5 | 20 | 80 | 1 |
Table 1. Gradient elution profile for urine samples.
The resolution between As(III) and DMA is improved at higher temperatures at the expense of longer retention times for MMA and As(V). As(V) elutes at 2.5 minutes and a further 1-minute re-equilibration time is included to re-establish initial conditions, giving a total run time of 3.5 minutes, which compares favorably with the CDC method run time of 17 minutes.
New methodology reduces chromatographic run times
Our methods are four to seven times faster than comparable separations previously reported, and can be very useful for increasing sample throughput for As speciation in response to changing regulations and increased need for biomonitoring.
Stay on top of the latest developments with the Agilent ICP-MS Journal
Are you interested in trace metals analysis and eager to keep up-to-date with the latest developments in the field of ICP-MS? Then take a closer look at Agilent’s dedicated ICP-MS Journal. Published four times a year and available as a PDF, you can view the latest issue of the ICP-MS Journal, plus all previous copies, by visiting the ICP-MS Journal Archive.
To receive a personal copy of the ICP-MS Journal direct to your inbox, please sign up here.
References
- C. P. Verdon, K. L. Caldwell, M. R. Fresquez and R. L. Jones, Determination of Seven Arsenic Compounds in Urine by
HPLC-ICPDRC-MS: A CDC Population Biomonitoring Method. Anal. Bioanal. Chem., 2009, 393(3), 939—947. - B. P. Jackson, Fast Ion Chromatography- ICP-QQQ for Arsenic Speciation. Journal of Analytical Atomic Spectrometry, 2015, 30, (6), 1405-1407.
Stay informed about the applications that are important to you
Subscribe to Access Agilent
Our free customized
monthly eNewsletter
Article Directory – April 2016
All articles in this issue
 Analyze low-ppm levels of active sulfur compounds with inert sample path on the Agilent 490 Micro GC
Analyze low-ppm levels of active sulfur compounds with inert sample path on the Agilent 490 Micro GC Fast arsenic speciation for food and urine analysis with Agilent LC-ICP-QQQ
Fast arsenic speciation for food and urine analysis with Agilent LC-ICP-QQQ Simplify achiral-chiral analysis of warfarin metabolites using Agilent 2D-LC/MS solutions
Simplify achiral-chiral analysis of warfarin metabolites using Agilent 2D-LC/MS solutions Optimized detection of gaseous sulfur compounds using Agilent J&W DB-Sulfur SCD GC column and Inert Flow Path
Optimized detection of gaseous sulfur compounds using Agilent J&W DB-Sulfur SCD GC column and Inert Flow Path Oil-free laboratory vacuum eliminates oil mess and disposal costs
Oil-free laboratory vacuum eliminates oil mess and disposal costs Eliminate conventional SEC roadblocks with Agilent AdvanceBio SEC columns
Eliminate conventional SEC roadblocks with Agilent AdvanceBio SEC columns Fast, economical assessment of herbals with Agilent 1290 Infinity LC system and LC columns
Fast, economical assessment of herbals with Agilent 1290 Infinity LC system and LC columns Agilent 5977B GC/MSD with HES offers improved VOC detection limits
Agilent 5977B GC/MSD with HES offers improved VOC detection limits
Figure 1

Isocratic separation of four arsenic species at 500 ng/L (ppt). 50 mM (NH4)2CO3 at 1.5 mL/min flow rate. Column temp. of 35 °C. As(V)* is the sum of As(V) and As(III) after oxidation.
Figure 2

Separation of six arsenic species spiked at 400 ng/L (ppt) in urine, using gradient conditions given in Table 1. Column temp. of 35 °C.