In environmental and food applications, doubly charged ion (M++) interferences arising from relatively high concentrations of rare earth elements (REEs) can affect the accurate measurement of arsenic (As) and selenium (Se).1 As shown in this foods study using the Agilent 8900 ICP-QQQ,2 most elements were measured in He KED mode, while As and Se were measured using oxygen (O2) cell gas, avoiding M++ interferences from 150Nd2+ and 150Sm2+ on 75As+, 156Gd2+ and 156Dy2+ on 78Se+.
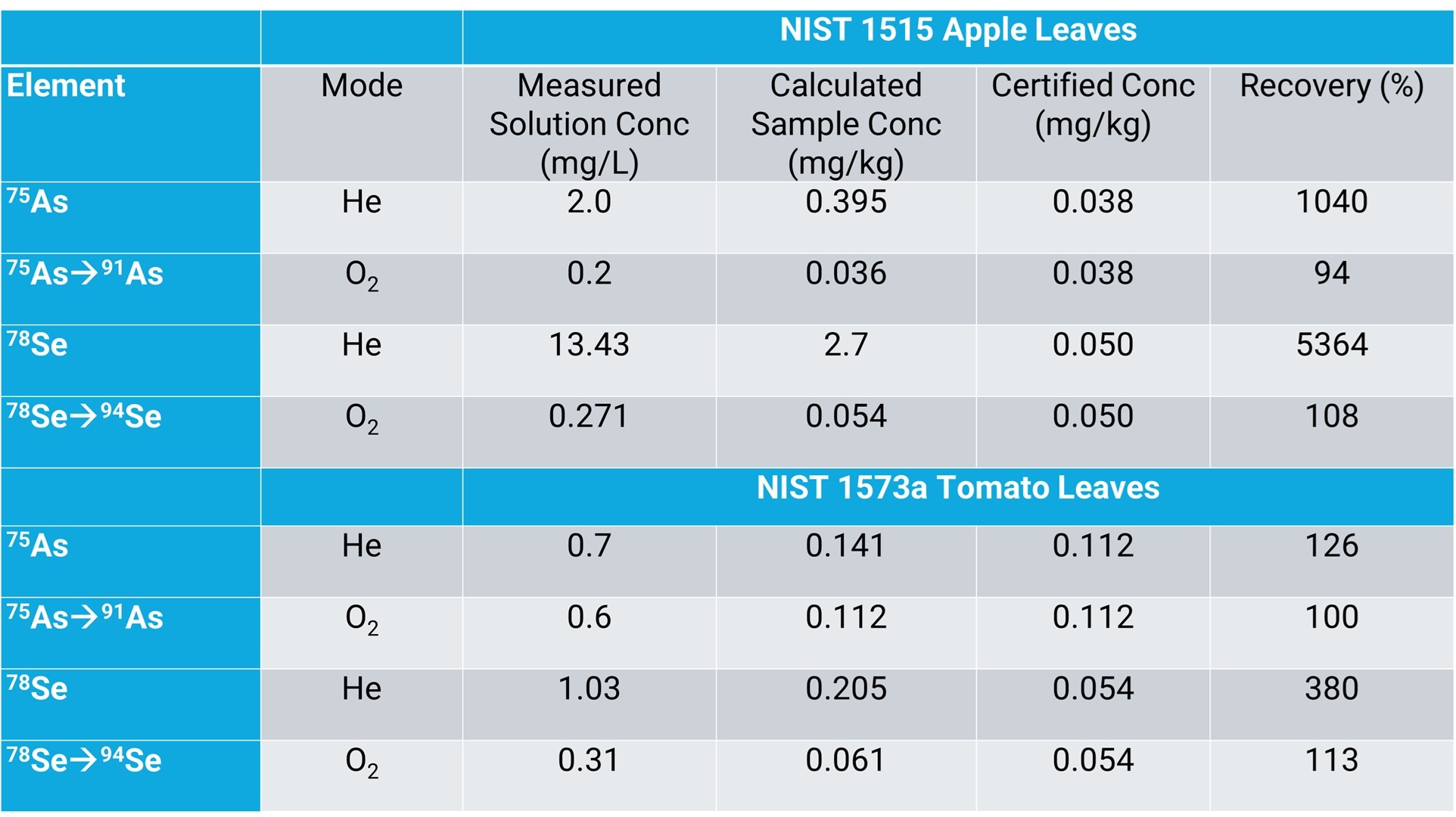
The accuracy of the method was evaluated by analyzing the five food certified reference materials (CRMs) as unknown samples. The comparison data in Table 1 highlight differences in the results for As and Se in a sample with a high level of REEs, depending on the method used (He or O2 mass-shift mode).
The NIST 1515 Apple Leaves CRM contains low μg/kg concentrations of As and Se and high concentrations of REEs. Reference (non-certified) values for Nd, Sm, and Gd are 17, 3, and 3 mg/kg, respectively. In the case of Apple Leaves (and Tomato Leaves), more accurate recovery was obtained for As and Se using O2 mass-shift mode, illustrating the potential error introduced by the relatively high level of REE in these reference materials.
In this environmental study, a similar approach was taken for the analysis of soils and sediments using the Agilent 8800 ICP-QQQ.3 REE++ interferences that can affect As and Se measurement at trace levels were avoided using O2 cell gas and MS/MS mass-shift mode. Most other elements were measured in He mode, which effectively removed common matrix-based polyatomic interferences in the complex and variable matrices of the samples.
Quantifying ultratrace analytes
As discussed in the ICP-MS Journal article on the analysis of basalt, single quadrupole ICP-MS supports trace level determination of REEs. For ultratrace applications, however, ICP-QQQ provides more reliable quantification due to its increased sensitivity, lower background, and more effective interference management capabilities.
In natural environmental waters, for example, the natural background concentration of the REEs is typically at or below the detection capabilities of conventional single quadrupole ICP-MS. Also, the analysis may be hindered by spectral interferences, such as BaO+ on Eu+, BaH+ on La+, or low-mass REE oxide ions overlapping high-mass REEs.
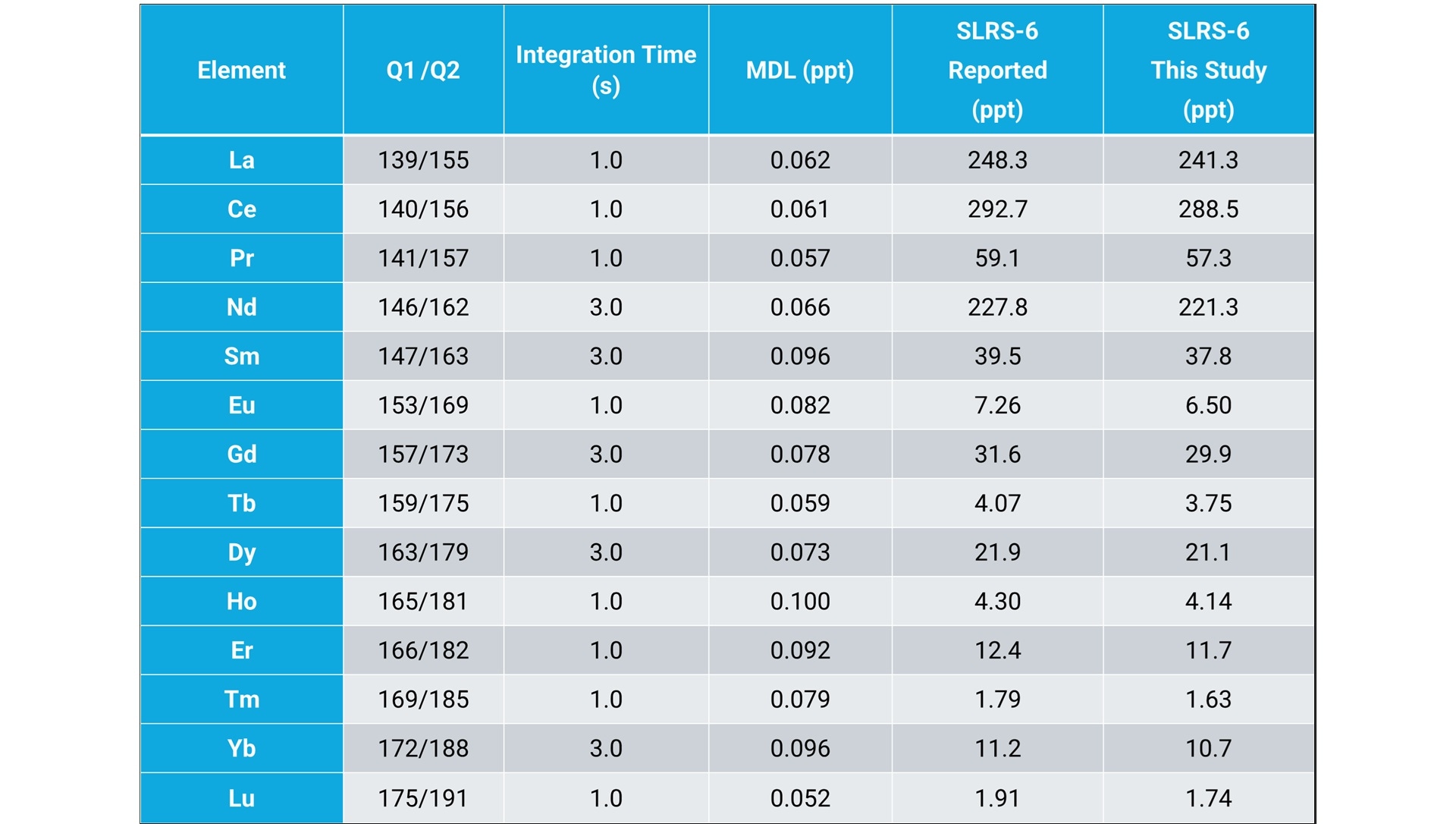
When the 8900 ICP-QQQ was used for the direct analysis of ultratrace REEs in river water, sub-ppt level method detection limits (MDLs) were achieved for the lanthanides (Table 2).4 The MS/MS method also showed good accuracy based on agreement between measured and certified elements in a river water CRM, SLRS-6 (NRC-CNRC, Ottawa, Canada).
Resolving isobaric spectral overlaps
ICP-QQQ can be used to improve the measurement of some less common contaminants in environmental samples, such as tellurium (Te). However, the only Te isotope free of isobaric overlap is 125Te, so it is the preferred isotope for ICP-MS measurements. But 125Te has an isotopic abundance of only 7.07%, which is almost a factor of five lower than the most abundant Te isotope.
To determine Te at lower levels in environmental samples, it would be beneficial to be able to measure the more abundant isotopes, 128Te (31.74%) and 130Te (34.08%). However, both isotopes suffer isobaric overlaps from Xe (a common trace contaminant in high-purity argon gas). In addition, the 130Te isotope is also overlapped by 130Ba, which may be present at mg/kg (ppm) levels in natural environmental samples. An ICP-MS/MS method that removes the Xe and Ba isobaric overlaps would significantly improve trace-level determination of Te.
In this study, isobaric overlaps from both Xe and Ba were chemically resolved from 128Te and 130Te using the 8900 ICP-QQQ and N2O and NH3 cell gases.5 The method allowed Te to be measured using its most abundant isotopes at m/z 128 and 130. Compared to no gas mode, the N2O/NH3 method reduced the contribution of Xe on 130Te by a factor of almost 2000 and ensured accurate analysis of Te in variable Ba matrices.
Solving oxide interferences: CoO+ on As+ and MoO+ on Cd+
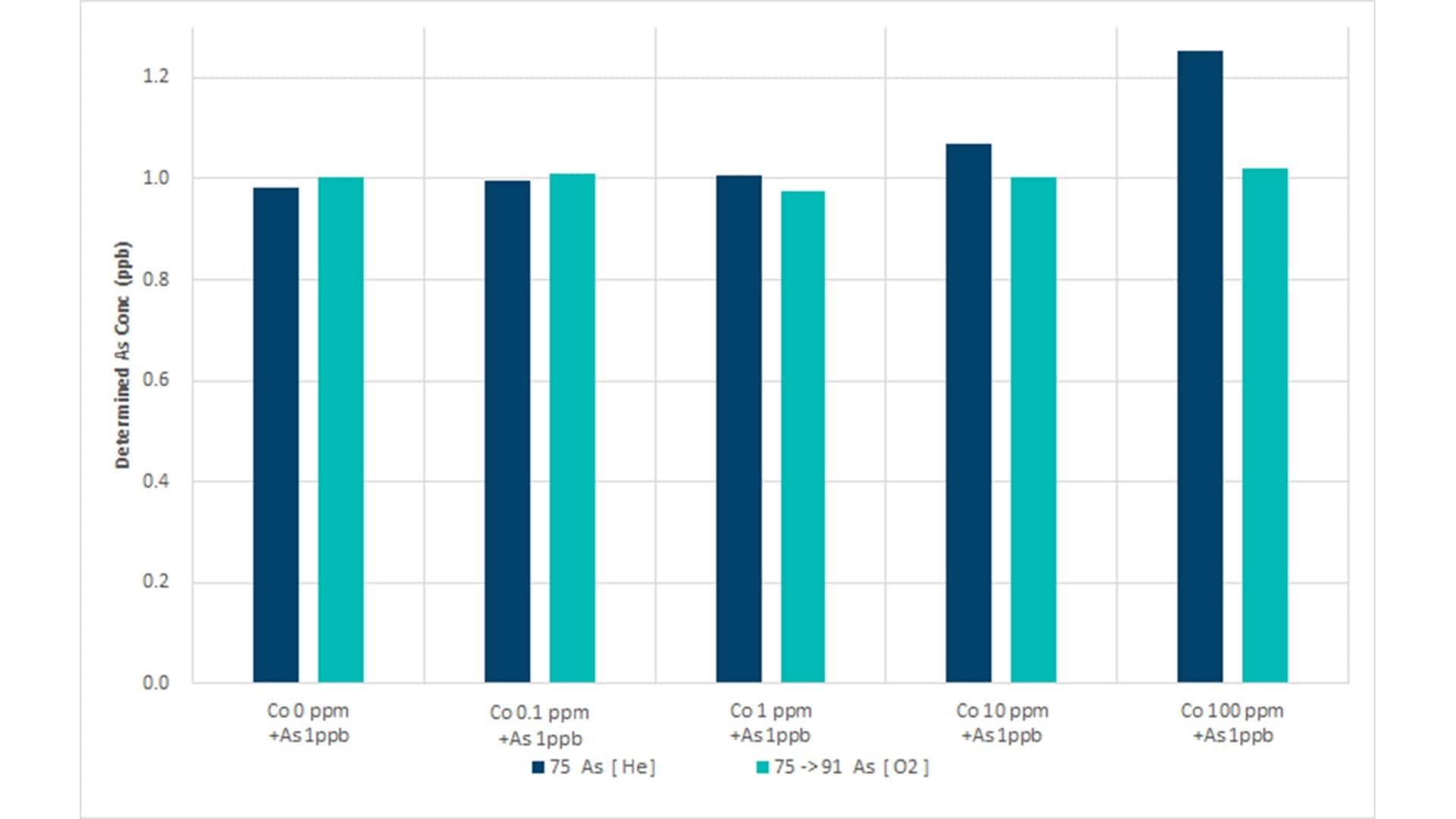
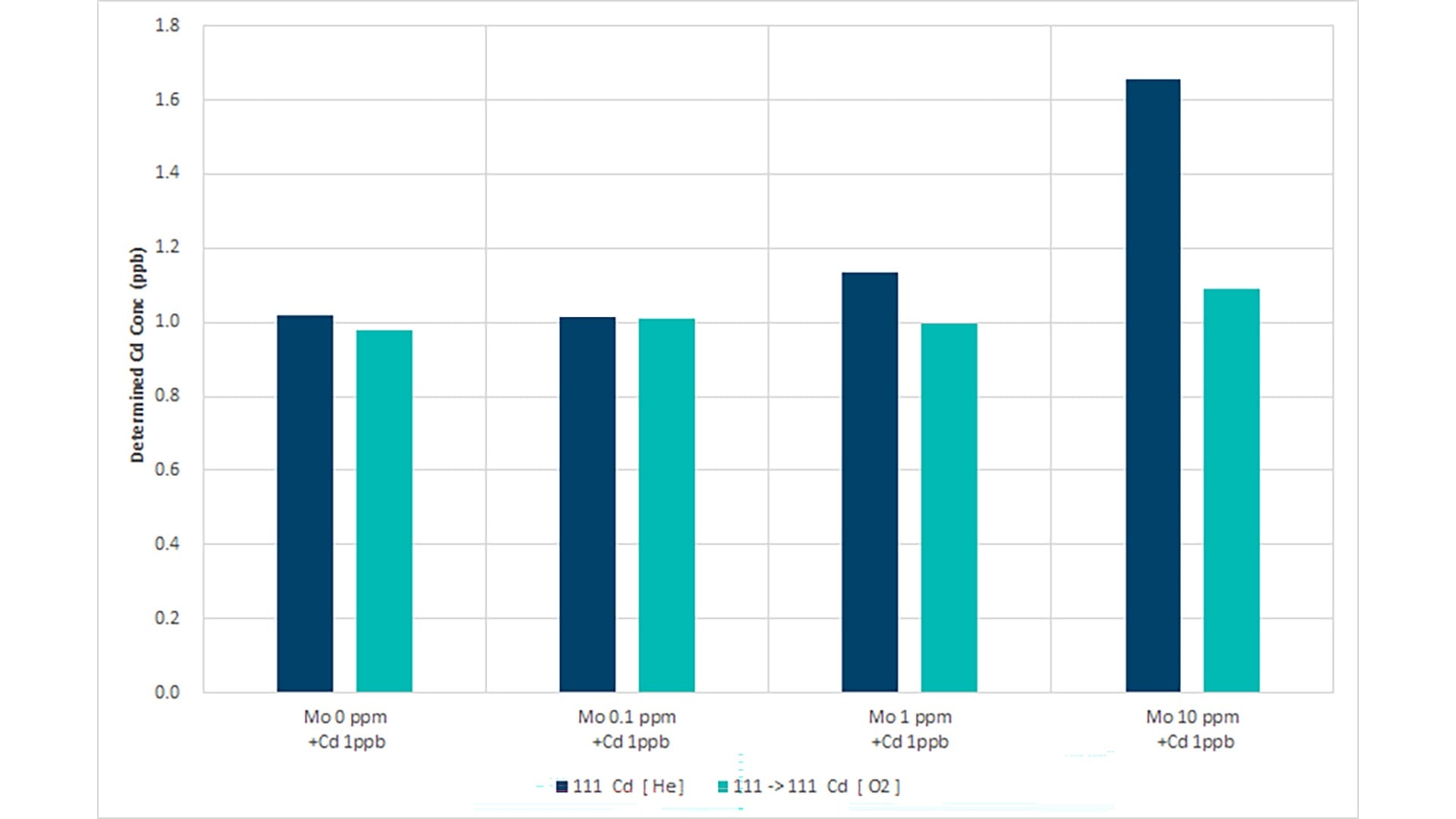
This study on the analysis of the heavy metals As, Cd, Hg, and Pb in dietary supplements shows that analysts can perform routine sample analyses without prior knowledge of potential interferences using the 8900 ICP-QQQ in MS/MS mode.6 Samples that contain a relatively high concentration of Co and Mo will form oxide-based interferences that impact the accurate measurement of As and Cd. He KED mode does not effectively eliminate oxide interferences such as 59Co16O+ on 75As+, 95Mo16O+ on 111Cd+, and 98Mo16O+ on 114Cd+, so an O2 mass-shift method was used.
CoO+ does not react with O2, while As+ reacts with O2 to form AsO, allowing AsO and Co to be separated. MoO+ reacts with O2 to form MoO2+, while Cd+ does not react with O2, allowing MoO and Cd to be separated.
As shown in Figures 1a and b, He mode is suitable for the analysis of As and Cd in matrices that contain Co and Mo at low ppm concentrations. However, poor recoveries were achieved for As in the presence of 100 ppm Co and Cd in a 10 ppm Mo matrix. In contrast, ICP-QQQ operating in MS/MS mode with O2 cell gas avoided oxide-ion interference for As and Cd at all matrix concentrations. The method allows consistent, low-level determination of As, measured as 75AsO+ at m/z 91, and 111Cd+ at m/z 111.
Tungsten-based interferences on mercury
Many mercury (Hg) compounds are toxic. So regulatory bodies, such as the US Food and Drug Administration (FDA), do not allow Hg in cosmetics, except under specific conditions when no other safe and effective preservatives are available.
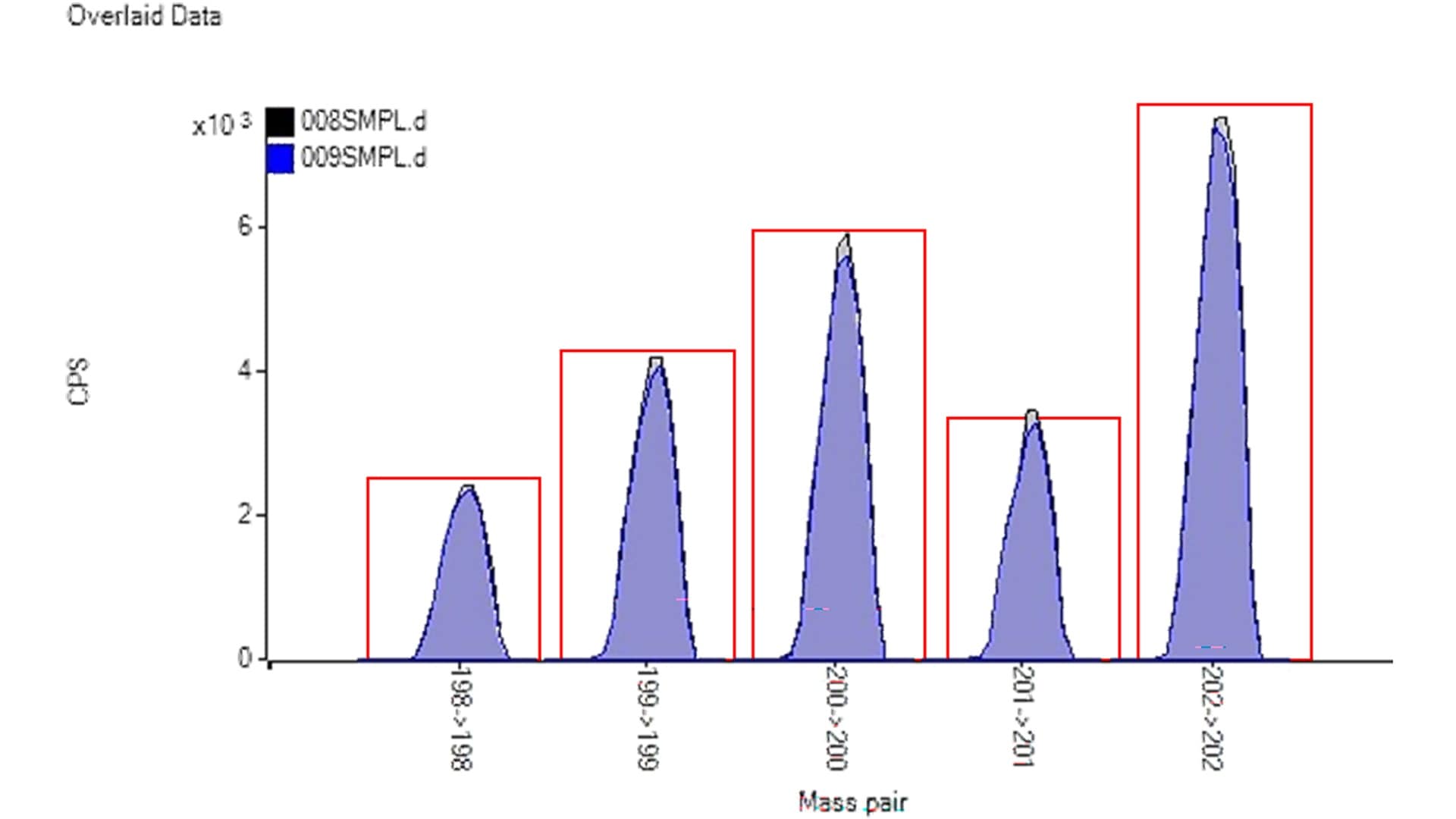
ICP-MS can be used successfully to perform trace-level analysis of Hg. However, some cosmetics contain large amounts of tungsten (W). This leads to polyatomic interferences from WO+ and WOH+ that affect all the Hg isotopes, making Hg measurement challenging. For example, the most abundant Hg isotopes, 200Hg and 202Hg, suffer overlaps from 184W16O+ and 186W16O+, respectively. Conventional single quadrupole ICP-MS is unable to resolve the WO+ and WOH+ interferences sufficiently to allow the accurate determination of Hg at trace levels. However, as shown in this study, the 8900 ICP-QQQ in O2 MS/MS on-mass mode can overcome these interferences, enabling the measurement of five major isotopes of mercury.7
A scan spectrum showing the mass range of the Hg isotopes was acquired for a 1 ug/L Hg standard (gray shading) and a solution containing the same concentration of Hg spiked into a high (10 mg/L) W matrix (purple shading). The overlaid spectra confirm that the measured isotopic abundances match the natural Hg isotope pattern despite the presence of the high W matrix in the second sample (Figure 2). A spike recovery test was performed by adding a 30 ppt Hg spike to a diluted cosmetic sample. Recovery was 104%, confirming the interference removal capability and matrix tolerance of the method.
ICP-QQQ as a routine analytical tool
We have highlighted only a few examples where Agilent ICP-QQQ operating in MS/MS mode resolves challenging spectral overlaps, enabling accurate quantification of even more elements in routine samples.
References
- Sugiyama, N. Solving Doubly Charged Ion Interferences using an Agilent 8900 ICP-QQQ
- Sakai, K. Benefits of the Agilent 8900 ICP-QQQ with MS/MS operation for routine food analysis
- Sakai, K. Routine soil analysis using an Agilent 8800 ICP-QQQ
- Sugiyama, N. Direct Analysis of Ultratrace Rare Earth Elements in Environmental Waters by ICP-QQQ
- Woods, G.; McCurdy, E. Using Double Mass Selection and Reaction Cell Gases to Resolve Isobaric Spectral Overlaps in ICP-MS
- Sakai, K. Routine Elemental Analysis of Dietary Supplements using an Agilent 8900 ICP-QQQ
- Wang, X; Wang, K; et al. Accurate Analysis of Trace Mercury in Cosmetics using the Agilent 8900 ICP-QQQ
