Access Agilent eNewsletter July 2016

Quick, accurate, cost-effective measurements of veterinary drugs in meat with Agilent MS/MS, UHPLC, and Q-TOF
Tarun Anumol, Agilent LC/MS Applications Scientist
Veterinary drugs are used in livestock to prevent disease and promote growth. Excessive use can result in contamination of meat, therefore measuring for veterinary drug residues in food is essential. Foods of animal origin, such as meat, are particularly challenging for analytical chemists to investigate effectively due to their complex nature.
Comprehensive solution provides accurate measurement and monitoring
Successful measurement of veterinary drugs typically requires sophisticated analytic methods and instruments, often based on LC/MS/MS technology. In this study, a 12-minute analytical method was developed for 122 veterinary drugs in meat. Agilent used its All Ions MS/MS on an Agilent 6545 Q-TOF LC/MS along with the Agilent veterinary drugs personal compound database and library (PCDL).
Agilent Q-TOF provides fast, sensitive data collection
All 122 veterinary drugs used in this analysis were spiked into bovine liver, kidney, and muscle tissue at levels of 0.5, 1.0, and 2.0 times the maximum tolerance levels for each drug that were determined by the USDA.
The spikes were analyzed using the Agilent All Ions data acquisition mode in an Agilent 6545 Q-TOF LC/MS. Additionally, the Agilent MassHunter Qualitative software was used to detect and verify the presence of these compounds. The PCDL provides MS/MS spectral and retention time information about each compound, enabling a data review process to quickly and reliably filter out false positives. At all three spike levels, more than 92% of veterinary drugs were detected in every matrix using the 6545 Q-TOF LC/MS.
Agilent 1290 Infinity UHPLC delivers lowest dispersion and highest resolution
An Agilent 1290 Infinity ultrahigh-performance liquid chromatograph (UHPLC) with a 40 µL loop HiPALS autosampler was used for this method. Separations were performed with an Agilent ZORBAX Eclipse Plus C-18 column (2.1 × 100 mm) using a gradient of water +0.1% formic acid (A) and MeCN +0.1% formic acid (B). To protect and enhance column lifetime, an Agilent inline filter was installed after use of the autosampler with an Agilent ZORBAX Eclipse plus guard column (2.1 × 5 mm) before the actual analytical column. Figure 1 illustrates a sample chromatogram of a veterinary drug standard at 50ng/mL in 80/20 water/MeCN.

Figure 2. A) Mass spectra and isotope spacing of Novobiocin in muscle. B) Library spectra in Agilent PCDL at CE: 10 eV Novobiocin with precursor (I) and three most abundant fragments (II, III, and IV), compared to (C) actual sample data. Compound positively identified.
Identifying veterinary drugs in meat with All Ions MS/MS
The Agilent All Ions MS/MS workflow identifies precursor masses and uses the spectral data available in the Agilent PCDL to look for coeluting fragments in the high collision energy channel. Figure 2 illustrates the component pieces of data that are used to automatically verify the results with Agilent MassHunter Qualitative software and the All Ions MS/MS approach.
Figure 2A (inset) indicates the mass spectrum for novobiocin identified in bovine muscle at the 1X tolerance level with the expected isotope abundancies and spacing in red boxes matched up against actual data (vertical red sticks). The mass accuracy, isotope spacing, isotope abundance, and retention time (RT) matching with the PCDL accounted for a total score of 98.41. Figure 2B (inset) is the MS/MS spectrum for novobiocin at CE of 10 V, available in the Agilent Veterinary Drugs PCDL, with the [M+H]+ precursor (I) and the most abundant fragment ions (II, III and IV) denoted. Figure 2C (inset) shows the actual chromatographic peaks seen in the muscle sample.
High-resolution data analysis achieved with Agilent MassHunter software
The All Ions MS/MS workflow is designed to acquire high-resolution MS data simultaneously in low and high collision energy channels in order to collect molecular and fragment ion data in the same run. Data acquisition was performed using Agilent MassHunter software, while Agilent MassHunter Qualitative software was used for data analysis. The Find by Formula feature in Agilent MassHunter Qualitative Analysis was used with the database search function to take advantage of the PCDL information.
The PCDL consisted of all the veterinary drugs necessary for this study with information on molecular formulas, exact monoisotopic mass, CAS number, MS/MS spectra collected at 0, 10, 20, and 40V for the [M+H]+ ion and retention times obtained from running standards against the developed LC method. This allowed significant increase in specificity of identification. Figure 3 illustrates the All Ions MS/MS workflow, including data processing filters used for the analysis of veterinary drugs in this study.
Integrated Agilent solution delivers superior detection, separation, and analysis of veterinary drugs in meat
At its conclusion, this experiment demonstrated the ability of the Agilent 1290 Infinity UHPLC with its unmatched separation and detection performance and the Agilent 6545 Q-TOF LC/MS with its high resolution and ultra sensitivity to analyze over 120 veterinary drugs in relevant matrices, including bovine muscle, liver, and kidney at ng/ng levels.
The use of a simple and efficient Agilent All Ions MS/MS workflow allows for analyte detection and identification using fragment ions in the same analytical run. This dramatically reduces potential false positives. The ability to perform quantification was demonstrated with calibration curve generation. As a result, sensitive qualitative and quantitative information for veterinary drugs in meat can be performed using the Agilent 6545 Q-TOF LC/MS.
For more information on this application, please explore Agilent publication 5991-6651EN.
Agilent offers a huge selection of solutions for data collection and analysis
Agilent offers a broad array of liquid chromatography, mass spectrometry, and LC/MS solutions, as well as related software and services, that can increase throughputs and efficiencies in your lab. Contact an Agilent representative today to learn more.
Stay informed about the applications that are important to you
Subscribe to Access Agilent
Our free customized
monthly eNewsletter
All articles in this issue
 Make your Q-TOF LC/MS analyses faster, easier, and more productive—regardless of your application
Make your Q-TOF LC/MS analyses faster, easier, and more productive—regardless of your application Agilent J&W DB-624UI capillary GC column excels for challenging applications with active compounds
Agilent J&W DB-624UI capillary GC column excels for challenging applications with active compounds Tip: Using enzymes in dissolution testing of gelatin capsules
Tip: Using enzymes in dissolution testing of gelatin capsules InfinityLab Poroshell 120 columns maximize LC workflow efficiency
InfinityLab Poroshell 120 columns maximize LC workflow efficiency Emerging life science applications of FTIR Imaging: Providing spatially resolved molecular information in disease research
Emerging life science applications of FTIR Imaging: Providing spatially resolved molecular information in disease research New LC/TQ database and method for central carbon metabolites
New LC/TQ database and method for central carbon metabolites Agilent QQQ systems enable ground-breaking research on use of metabolite biomarkers to predict a common pregnancy complication
Agilent QQQ systems enable ground-breaking research on use of metabolite biomarkers to predict a common pregnancy complication Analysis of PEGylated proteins with Agilent AdvanceBio SEC columns
Analysis of PEGylated proteins with Agilent AdvanceBio SEC columns Structural elucidation of in-process impurities using high resolution LC/MS/MS
Structural elucidation of in-process impurities using high resolution LC/MS/MS Removal of lipids for the analysis of toxicological compounds in plasma by LC/MS/MS
Removal of lipids for the analysis of toxicological compounds in plasma by LC/MS/MS Quick, accurate, cost-effective measurements of veterinary drugs in meat with Agilent MS/MS, UHPLC, and Q-TOF
Quick, accurate, cost-effective measurements of veterinary drugs in meat with Agilent MS/MS, UHPLC, and Q-TOF
Figure 1
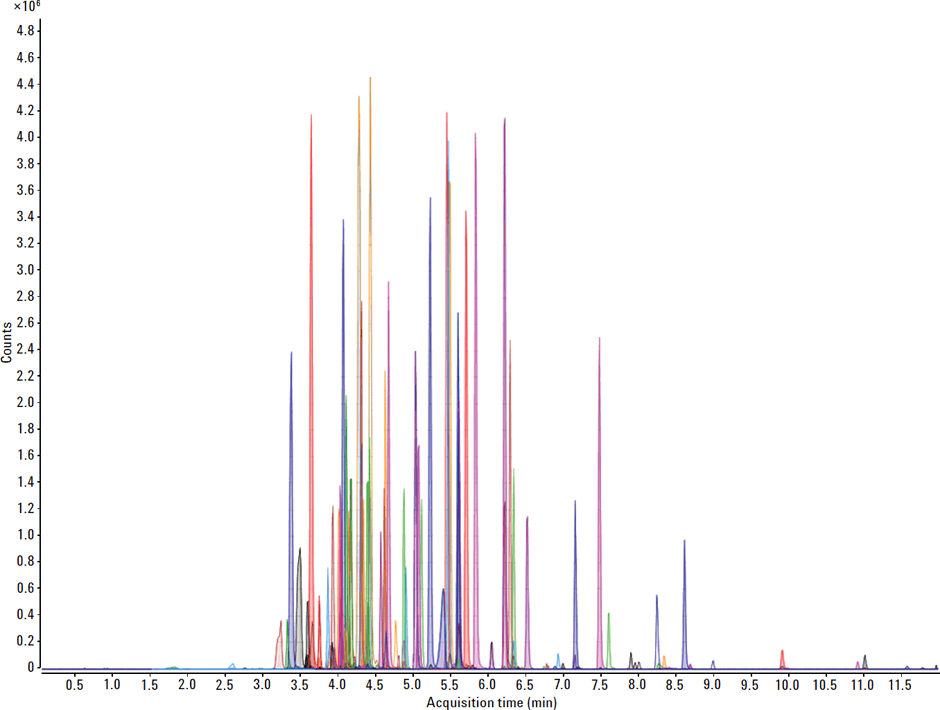
Sample chromatogram of a VD standard at 50 ng/mL in 80/20 water/MeCN.
Figure 2

A) Mass spectra and isotope spacing of Novobiocin in muscle. B) Library spectra in Agilent PCDL at CE: 10 eV Novobiocin with precursor (I) and three most abundant fragments (II, III, and IV), compared to (C) actual sample data. Compound positively identified.
Figure 3

Agilent All Ions MS/MS data analysis workflow.