Access Agilent eNewsletter July 2016
Removal of lipids for the analysis of toxicological compounds in plasma by LC/MS/MS
Joan Stevens, Agilent Senior Applications Scientist
A convenient analytical method for the determination of toxicological compounds in plasma involves the addition of acetonitrile to a small volume of plasma to elicit protein precipitation. The supernatant is transferred to a dispersive tube containing Enhanced Matrix Removal—Lipid (EMR–Lipid) sorbent to remove > 97% endogenous plasma lipid matrix components. Compared to competitors, Agilent EMR–Lipid removed substantially more lipids than other phospholipid removing sorbents tested. Analytes are isolated from spiked plasma samples with accuracies above 95% and Relative Standard Deviations (RSDs) < 6% on average.
Combining protein precipitation with EMR–Lipid dispersive SPE for plasma samples offered separation for various toxicological compounds and Limits of Quantification (LOQs) at 1 ng/mL or below based on method performance. The method is quick, easy, and removes lipids that are known to remain on the column causing chromatographic anomalies and source contamination.
Protein precipitation and cleanup of toxicologically monitored compounds
The experiments presented in this application used plasma containing NaEDTA or NaCitrate, anticoagulant agents. Protein precipitation was performed with acidified acetonitrile, followed by cleanup with Agilent EMR–Lipid and enhanced post sample treatment (polish and dry step). The experiments were performed using 25 different toxicologically monitored compounds with a broad range of hydrophobicity and pKa (Table1).
| Compound | CAS number | Log P | pKa |
|---|---|---|---|
| Alprazolam | 028981-97-7 | 3.02 | 1.79, 5.08 |
| Amphetamine | 000051-63-8 | 1.80 | 10.01 |
| Cocaine | 000053-21-4 | 2.28 | 8.85 |
| Codeine | 000076-57-3 | 1.34 | 9.19 |
| Diazepam | 000439-14-5 | 3.08 | 2.92 |
| Heroin | 005893-91-4 | 1.55 | 9.10 |
| Hydrocodone | 000143-71-5 | 1.96 | 8.61 |
| Lorazepam | 000846-49-1 | 3.53 | 12.46 |
| MDA 3,4-methylenedioxyamphetamine hydrochloride | 013673-99-9 | 1.43 | 10.01 |
| MDEA 3,4-methylenedioxyl-N-ethyl-amphetamine hydrochloride | 116261-63-3 | 2.22 | 10.22 |
| MDMA 3,4-methylenedioxymethamphetamine hydrochloride | 092279-84-0 | 1.86 | 10.14 |
| Meperidine hydrochloride | 000050-13-5 | 2.46 | 8.16 |
| Methadone | 001095-90-5 | 5.01 | 9.12 |
| Methamphetamine hydrochloride | 000051-57-0 | 2.24 | 10.21 |
| Nitrazepam | 000146-22-5 | 2.89 | 2.60, 3.28 |
| Oxazepam | 000604-75-1 | 2.92 | 10.61, 12.47 |
| Oxycodone | 000124-90-3 | 1.03 | 8.21 |
| PCP Phencyclidine hydrochloride | 000956-90-1 | 4.49 | 10.64 |
| Phentermine | 000122-09-8 | 2.08 | 10.25 |
| Proadifen | 000062-68-0 | 5.61 | 8.96 |
| Strychnine | 000057-24-9 | 0.93 | 9.27 |
| Temazepam | 000846-50-4 | 2.79 | 10.68 |
| THC | 001972-08-3 | 5.94 | 9.34 |
| Trazodone | 025332-39-2 | 3.13 | 7.09 |
| Verapamil | 000152-11-4 | 5.04 | 9.68 |
Table 1. EMR-Lipid dispersive SPE cleanup of 25 different toxicologically monitored compounds with a broad range of hydrophobicity and pKa.
Maintaining optimal chromatography with cleaner matrix
Protein precipitation is a commonly used sample preparation technique with biological samples. Although it does remove proteins from the extract, lipid coextraction with target analytes can cause chromatographic anomalies and MS ion suppression. Agilent Bond Elut EMR–Lipid selectively removes lipids through size exclusion and hydrophobic interactions without affecting analyte recovery. Phospholipids were monitored using LC/MS/MS for product ion m/z 184 and MS1 from 100 to 1000. The chromatographic runtime was extended out to 30 minutes to elute matrix coextractives remaining on the column and possibly in the ion source. These coextractives would not necessarily be apparent in a short fast gradient used for analyte analysis, but can elute in subsequent injections, causing analytical variability.
Efficient lipid removal with Agilent EMR
To compare the lipid removal efficiency of Agilent EMR–Lipid to other commercially available phospholipid removing sorbents, an aliquot of 500 µL of plasma was mixed with 1 mL of ACN (0.2 % FA) to precipitate the proteins and then passed through the phospholipid cartridges by vacuum. Figure 1 demonstrates the remarkably efficient lipid cleanup of Agilent EMR–Lipid when compared to common phospholipid removing sorbents; Waters Oasis Prime, Phenomenex Phree, and Supleco HybridSPE. Figure 3 is the representative chromatographic overlay of the dynamic MRM for a 10 ng/mL spike plasma sample after protein precipitation and EMR–Lipid dispersive SPE cleanup.
Higher accuracy and easier reproducibility with Agilent EMR—Lipid
The accuracy and reproducibility was determined by spiking standards into the plasma samples at 5, 25, and 50 ng/mL, in replicates of five (n=5). These QC plasma samples were then subjected to protein precipitation and EMR–Lipid dispersive cleanup. Accuracy was determined against matrix matched calibration curve. The recovery and relative standard deviation are shown in Figure 2 and 3.
The results show excellent recoveries for all compounds with average recoveries at 5 ng/mL greater than 95% and RSD <6 %. Heroin was the only compound with relatively lower recoveries 65% on average, but RSDs below 8%.
Agilent miniaturized EMR—Lipid for optimized analysis of plasma
A simple and inexpensive sample clean-up method using protein precipitation and Agilent EMR–Lipid dispersive cleanup has been developed for plasma LC/MS/MS for a range of toxicological compounds. The advantages of this new method are: A cleaner extract with significant overall lipid removal compared to standard PPT and commercially available lipid removal products. Excellent recoveries and single digit % RSDs. Substantially cleaner extracts without the need for extra instrumentation or glassware, offer an easy and user-friendly sample preparation. This approach for the extraction of toxicological compounds from plasma is easily implemented into laboratories without extensive expertise in sample preparation techniques since it only involves spiking, vortexing and centrifugation.
Agilent provides a wide array of solutions and products for effective sample prep
Agilent’s sample preparation products help you to achieve your analytical goals. The application discussed in this article utilized the Agilent Bond Elut Enhanced Matrix Removal—Lipid technique for analysis of toxicological compounds in plasma. We demonstrated effective extraction of compounds from biological matrices, as well as superior sensitivity with LOQs at or below 1 ng/mL. For more information on the innovative EMR—Lipid methodology, please contact an Agilent representative today.
For Research Use Only. Not for use in diagnostic procedures.
This information is subject to change without notice.
Stay informed about the applications that are important to you
Subscribe to Access Agilent
Our free customized
monthly eNewsletter
All articles in this issue
 Make your Q-TOF LC/MS analyses faster, easier, and more productive—regardless of your application
Make your Q-TOF LC/MS analyses faster, easier, and more productive—regardless of your application Agilent J&W DB-624UI capillary GC column excels for challenging applications with active compounds
Agilent J&W DB-624UI capillary GC column excels for challenging applications with active compounds Tip: Using enzymes in dissolution testing of gelatin capsules
Tip: Using enzymes in dissolution testing of gelatin capsules InfinityLab Poroshell 120 columns maximize LC workflow efficiency
InfinityLab Poroshell 120 columns maximize LC workflow efficiency Emerging life science applications of FTIR Imaging: Providing spatially resolved molecular information in disease research
Emerging life science applications of FTIR Imaging: Providing spatially resolved molecular information in disease research New LC/TQ database and method for central carbon metabolites
New LC/TQ database and method for central carbon metabolites Agilent QQQ systems enable ground-breaking research on use of metabolite biomarkers to predict a common pregnancy complication
Agilent QQQ systems enable ground-breaking research on use of metabolite biomarkers to predict a common pregnancy complication Analysis of PEGylated proteins with Agilent AdvanceBio SEC columns
Analysis of PEGylated proteins with Agilent AdvanceBio SEC columns Structural elucidation of in-process impurities using high resolution LC/MS/MS
Structural elucidation of in-process impurities using high resolution LC/MS/MS Removal of lipids for the analysis of toxicological compounds in plasma by LC/MS/MS
Removal of lipids for the analysis of toxicological compounds in plasma by LC/MS/MS Quick, accurate, cost-effective measurements of veterinary drugs in meat with Agilent MS/MS, UHPLC, and Q-TOF
Quick, accurate, cost-effective measurements of veterinary drugs in meat with Agilent MS/MS, UHPLC, and Q-TOF
Figure 1
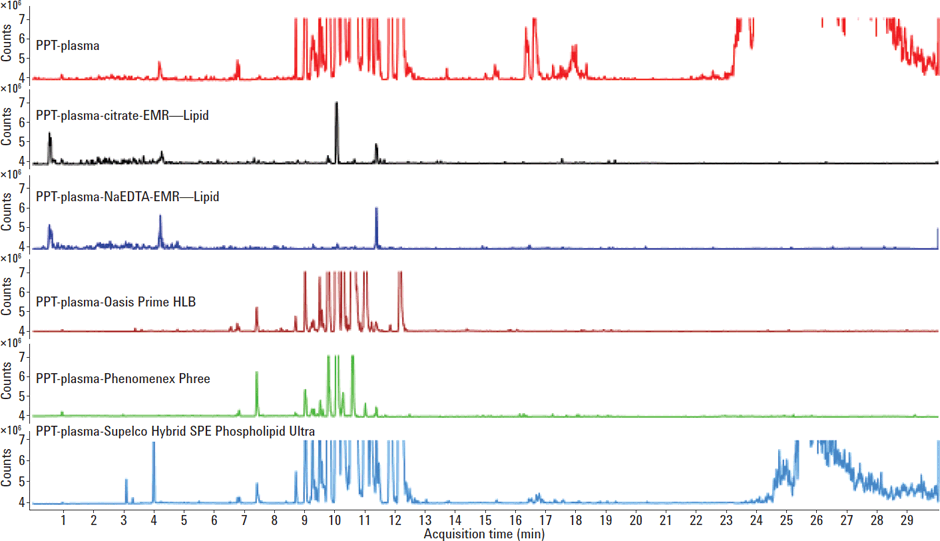
LC/MS/MS product ion m/z 184 for plasma sample after protein precipitation and various lipid removal sorbents.
Figure 2
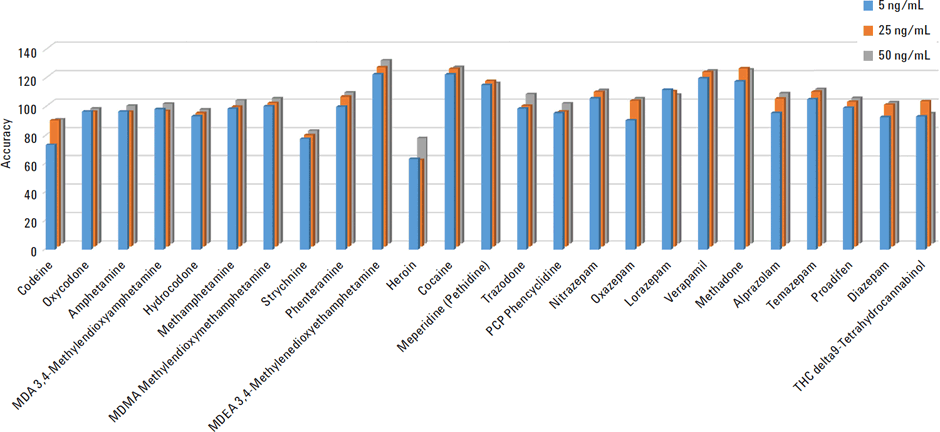
Accuracy for the 25 extracted toxicological compounds from human plasma.
Figure 3
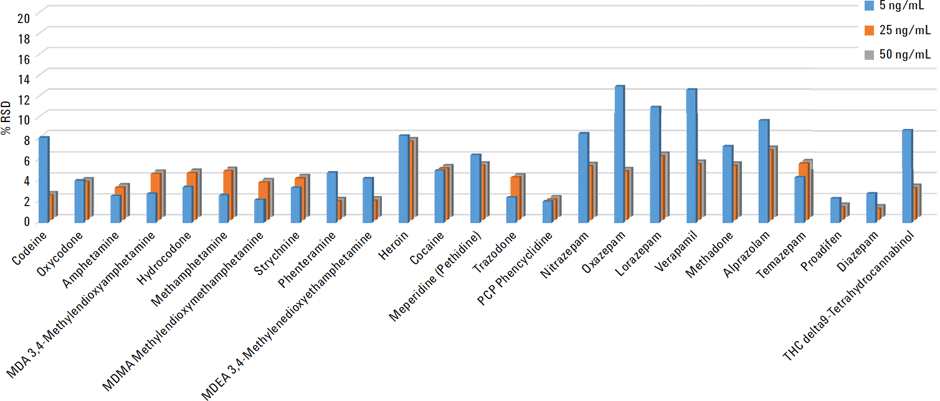
Relative Standard Deviation (RSDs) for the 25 toxicological compounds in human plasma.