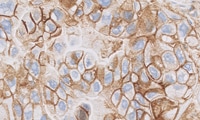
PD-L1 is a proven biomarker for patient response to KEYTRUDA in urothelial carcinoma1,2
- More than a third of all global incidences of urothelial carcinoma are reported in Europe, and few advancements in treatment have been made
- Anti-PD-1 therapies are a promising new treatment class for many cancer types—the KEYNOTE-052 and KEYNOTE-045 clinical trials studied the efficacy of KEYTRUDA, an anti-PD-1 monotherapy, at first-line and second-line respectively, in specific urothelial carcinoma patient populations
- IHC testing for PD-L1 expression levels enables identification of patients most likely to benefit from anti-PD-1 monotherapy
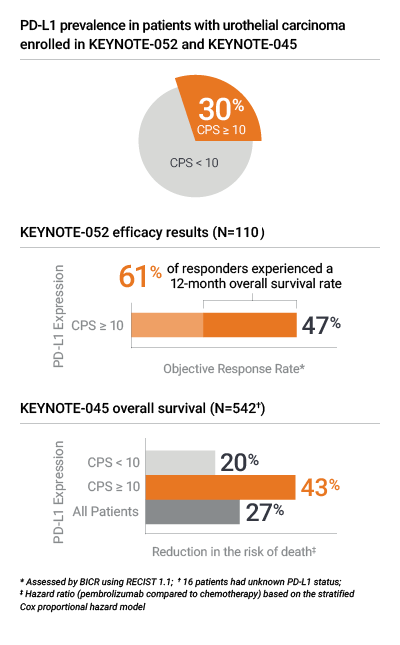
Proven in KEYNOTE-052 and KEYNOTE-045 for PD-L1 results in urothelial carcinoma1,2
- PD-L1 IHC 22C3 pharmDx was the only IHC assay used to assess PD-L1 expression in the KEYNOTE-052 and KEYNOTE-045 trials
- KEYNOTE-052 evaluated objective response rate (ORR) in locally advanced or metastatic urothelial carcinoma patients ineligible for cisplatin-containing chemotherapy
- KEYNOTE-045 evaluated overall survival in locally advanced or metastatic urothelial carcinoma patients previously treated with platinum-based chemotherapy

More than half of all patients with advanced urothelial carcinoma are ineligible for first-line cisplatin-based chemotherapy treatment
- Renal dysfunction, poor performance status, or other comorbidities can cause patients to be ineligible for treatment
- There is no standard of care for patients who can not receive cisplatin-based chemotherapy
- Additionally, for patients who receive cisplatin-based chemotherapy and relapse, there is no standard of care and the median overall survival is reduced to 6–7 months



KEYTRUDA® is a registered trademark of Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA
References: 1. PD-L1 IHC 22C3 pharmDx [package insert]. Carpinteria, CA: Dako, Agilent Pathology Solutions; 2019. 2. Keytruda [package insert]. Hoddesdon, UK: Merck Sharp & Dohme Limited; 2019. 3. Antoni S, Ferlay J, Soerjomataram I, Znaor A, Jemal A, Bray F. Bladder cancer incidence and mortality: A global overview and recent trends. Eur Urol. 2017;71(1):96-108. 4. Choueiri TK, Ross RW, Jacobus S, et al. Double-blind, randomized trial of docetaxel plus vandetanib versus docetaxel plus placebo in platinum-pretreated metastatic urothelial cancer. J Clin Oncol. 2012;30(5):507-512. 5. Bellmunt J, Mottet N, De Santis M. Urothelial carcinoma management in elderly or unfit patients. EJC Suppl. 2016;14(1):1-20. 6. Balar AV, Castellano D, O'Donnell PH, et al. First-line pembrolizumab in cisplatin-ineligible patients with locally advanced and unresectable or metastatic urothelial cancer (KEYNOTE-052): A multicentre, single-arm, phase 2 study. Lancet. 2017;18(11):1483-1492. 7. Galsky MD, Hahn NM, Rosenberg J, et al. Treatment of patients with metastatic urothelial cancer "unfit" for cisplatin-based chemotherapy. J Clin Oncol. 2011;29(17):2432-2438. 8. Bellmunt J, de Wit R, Vaughn DJ, et al. Pembrolizumab as second-line therapy for advanced urothelial carcinoma. N Engl J Med. 2017;376(11):1015-1026.
For countries outside of the European Union, see the local KEYTRUDA product label for approved indications and expression cutoff values to guide therapy.
D49802/02.1