Access Agilent eNewsletter, November 2014
>> Update My Profile | Subscribe to Access Agilent | Article Directory

New Agilent biocolumn analyzes mAbs with increased speed, resolution, recovery, efficiency, and reproducibility
By Wu Chen
Agilent R&D Senior Chemist
Anne Mack
Agilent BioHPLC Applications Chemist
and Stephen Luke
Agilent LC Columns Product Manager
Monoclonal antibody (mAb) therapeutics have been a part of several major news stories this year, most notably for the use of an experimental ZMapp three-antibody cocktail to create an artificial immune response against sugar-tagged proteins on the outside of the Ebola virus.
High Performance Liquid Chromatography (HPLC) and Ultra High Performance Liquid Chromatography (UHPLC) systems are commonly used for biopharmaceutical discovery and development of mAbs. To drive progress in this exciting arena, Agilent developed a new family of AdvanceBio RP-mAb LC columns designed to increase the speed, resolution, recovery, and efficiency of analyzing intact and reduced monoclonal antibodies.
AdvanceBio RP-mAb columns are based on Agilent Poroshell technology, featuring superficially porous 3.5 µm silica particles with a 450Å pore diameter, the largest pore size available for superficially porous particles in protein and mAb separations. The 0.25 µm porous layer is formed in a single step instead of a multi-layer process, which provides highly uniform particles with excellent batch-to-batch reproducibility. Specifically designed for reversed-phase separations of intact and reduced monoclonal antibodies, this unique particle is available with StableBond C8, C4, and Diphenyl bonding chemistries to provide real resolution power.
 Enlarge
Enlarge
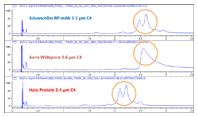
Figure 1. Higher resolution for IgG2 with the Agilent AdvanceBio RP-mAb C4, 2.1 x 100 mm, 3.5 µm column.
 Enlarge
Enlarge
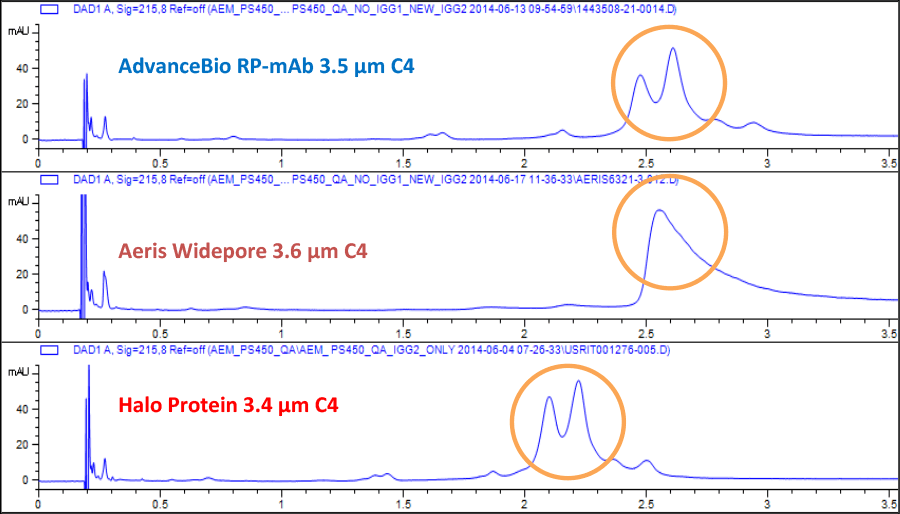
Figure 2. AdvanceBio RP-mAb produces taller, sharper, better-defined peaks when compared to a totally porous 300Ǻ column.
 Enlarge
Enlarge
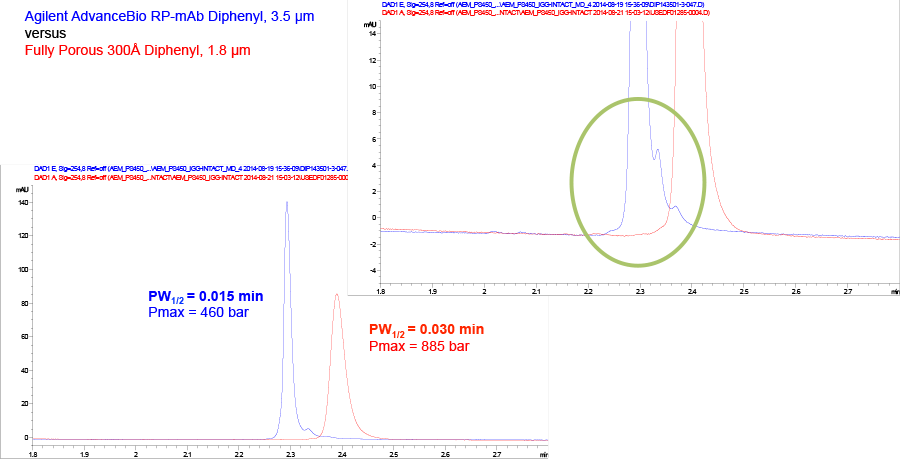
Figure 3. Agilent AdvanceBio RP-mAb C4, SB-C8, and Diphenyl phases provide different selectivity (conditions the same as Table 1).
Higher resolution with taller, sharper, better-defined peaks
Agilent AdvanceBio RP-mAb delivers higher resolution than columns designed for protein separation from other suppliers, as you can see in Figure 1.
In addition, our studies have demonstrated that AdvanceBio RP-mAb resolves more detail than 300Å totally porous columns. Figure 2 shows the taller, sharper, and more finely detailed peaks produced by AdvanceBio RP-mAb when compared to a totally porous particle column with a 300Å pore diameter.
Excellent reproducibility
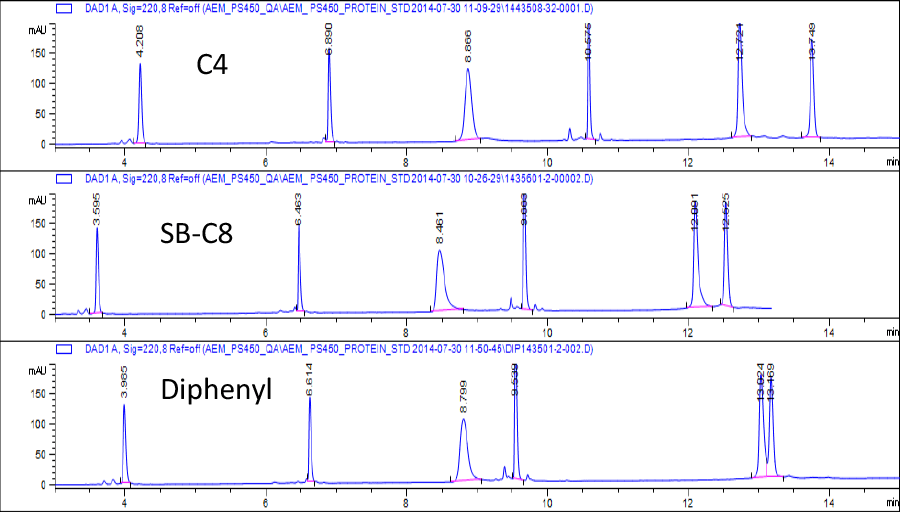
The Agilent Poroshell manufacturing process makes particles with first class reproducibility. In Table 1 the performance of protein separation with Agilent AdvanceBio RP-mAb 3.5 µm, C4 columns from ten different media batches is demonstrated. Note how the retention time and peak width are very reproducible over these columns, with %RSD less than 1.5% and 7.0%, respectively.
Protein |
%RSD |
|
|---|---|---|
Retention time (min) |
Peak width |
|
Ribonuclease A, 13.7 kDa |
1.20 |
3.14 |
Cytochrome C, 12 kDa |
0.80 |
3.90 |
Holo-transferrin, 80 kDa |
0.39 |
6.42 |
α-Lactalbumin, 14.2 kDa |
0.61 |
3.25 |
Catalase, 240 kDa |
0.32 |
4.19 |
Carbonic anhydrase, 29 kDa |
0.41 |
2.64 |
Table 1. %RSD of proteins of Agilent AdvanceBio RP-mAb 3.5 µm, C4 columns from ten different batches.
Selectivity options
Columns in the AdvanceBio RP-mAb range are available in three phases with different selectivities, giving you flexibility in method development to ensure the highest resolution for your application. Figure 3 shows how phase choice can tune the separation.
A robust family of high-quality HPLC and UHPLC solutions
AdvanceBio RP-mAb is the latest addition to Agilent’s growing family of high-quality AdvanceBio columns for characterizing monoclonal antibodies by HPLC and UHPLC. From sample purification to analysis, Agilent biomolecule columns are easy to integrate into your workflow for a complete, reproducible solution.
Agilent has a robust line of state-of-the-art products for all your liquid chromatography needs. These include the Agilent 1260 Infinity Bio-inert LC system with a metal-free sample path and the Agilent 1290 Infinity LC, designed to provide the highest speed, resolution, and ultra-sensitivity for UHPLC applications. While biomolecules are complex, analyzing their structure is simple and easy with Agilent HPLC columns, systems, and supplies. Why not take a few moments to explore these Biopharma solutions now? Then take advantage of our extensive Biopharma resources to maximize your lab’s investment in biopharmaceutical discovery and development of mAbs.
>> Update My Profile | Subscribe to Access Agilent | Article Directory
Figure 1.
Conditions |
|
|---|---|
Mobile phase |
A, 0.1% TFA in water; B, 0.1% TFA in acetonitrile |
Flow rate |
1.0 mL/min |
Gradient |
32 to 38% B in 8 min |
Temperature |
85 °C |
Injection |
5 µL of IgG2 Lambda (Sigma I5279-1MG) |
Detection |
215 nm |
Higher resolution for IgG2 with the Agilent AdvanceBio RP-mAb C4, 2.1 x 100 mm, 3.5 µm column.
Figure 2.
Conditions |
|
|---|---|
Columns |
2.1 x 100 mm |
Mobile phase |
A, 0.1% TFA in water:IPA (98:2), B, IPA:acetonitrile:M.P.A (70:20:10) |
Flow rate |
1.0 mL/min |
Gradient |
10 to 58% B in 4 min |
Temperature |
80 °C |
Injection |
5 µL of 1 mg/mL humanized recombinant Herceptin IgG1 intact (Creative Biolabs) |
AdvanceBio RP-mAb produces taller, sharper, better-defined peaks when compared to a totally porous 300Ǻ column.
Figure 3.
Conditions |
|
|---|---|
Columns |
2.1 x 100 mm |
Mobile phase |
A, 0.1% TFA in water, B: 0.1% TFA in acetonitrile |
Flow rate |
0.3 mL/min |
Gradient |
20 to 50% B in 15 min |
Temperature |
60 °C |
Detection |
220 nm |
Injection |
5 µL of of protein mix containing ribonuclease A, cytochrome C, holo-transferrin, α-lactalbumin, catalase, carbonic anhydrase |
Agilent AdvanceBio RP-mAb C4, SB-C8, and Diphenyl phases provide different selectivity (conditions the same as Table 1).