Access Agilent eNewsletter, September 2013
>> Update My Profile | Subscribe to Access Agilent | Article Directory

Ultrafast analyses of six antiepileptic drugs in serum with the Agilent RapidFire High-throughput Mass Spectrometry System
By Mohamed Youssef
Agilent Applications Scientist, RapidFire
and Vaughn P. Miller
Agilent Applications Manager, RapidFire
Traditional measurement methods for clinical research in quantitative antiepileptic drug analysis use HPLC, LC/MS/MS, and other technologies. The need for greater throughput and faster turn-around times has increased the demands on these traditional technologies. To solve the throughput problem, the Agilent RapidFire High-throughput Mass Spectrometry System delivers ultrafast solid phase extraction (SPE) MS/MS analyses with cycle times less than 15 seconds per sample.
 Enlarge
Enlarge
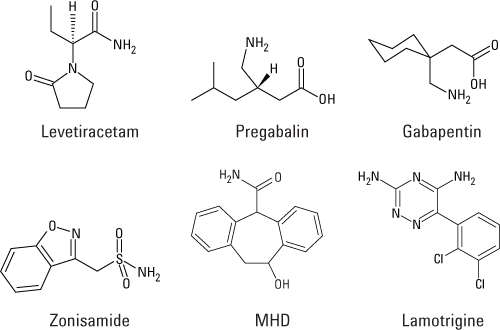
Figure 1. Chemical structures of the six antiepileptic drugs used to demonstrate throughput of more than four samples per minute.
We recently developed an ultrafast SPE/MS/MS method for simultaneous analyses in human serum of the six antiepileptic drugs (AEDs) shown in Figure 1. Compared with LC/MS/MS and HPLC assays, we achieved much faster sample cycle times and similar analytical results.
Amazing speed, accurate results
A simple protein precipitation methodology followed by dilute and shoot analysis by RapidFire SPE/MS/MS enables the accurate and precise measurement of these analytes in human serum over a linear range of 1 to 100 µg/mL. We analyzed samples on the RapidFire SPE/MS/MS system at an astounding 14 seconds per sample. This ultrafast method has the speed and accuracy necessary for an efficient quantitative workflow.
Our Agilent RapidFire/MS/MS system consisted of the following modules: Agilent RapidFire 300, Agilent 6460 Triple Quadrupole LC/MS System using Agilent MassHunter Workstation Triple Quadrupole Acquisition Software (B.04.01) with Qualitative Analysis (B.04.00), Quantitative Analysis (B.04.00), and RapidFire Acquisition Software.
We simultaneously monitored quantitative and qualitative ions for all six AEDs and internal standards in all experiments. Agilent MassHunter Workstation Quantitative Analysis automatically calculated qualifier ion ratios. Agilent Application Note 5991-2292EN provides the RapidFire/MS/MS conditions.
 Enlarge
Enlarge
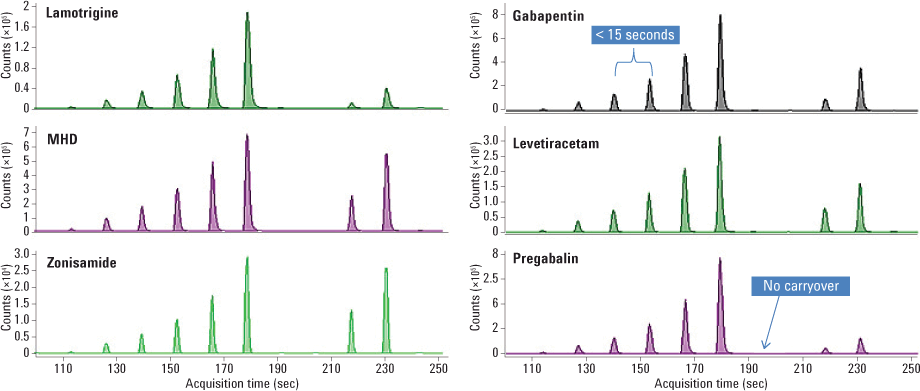
Figure 2. Representative calibration curves for each of the six AED analytes show the injection-to-injection interval of only 14 seconds.
 Enlarge
Enlarge
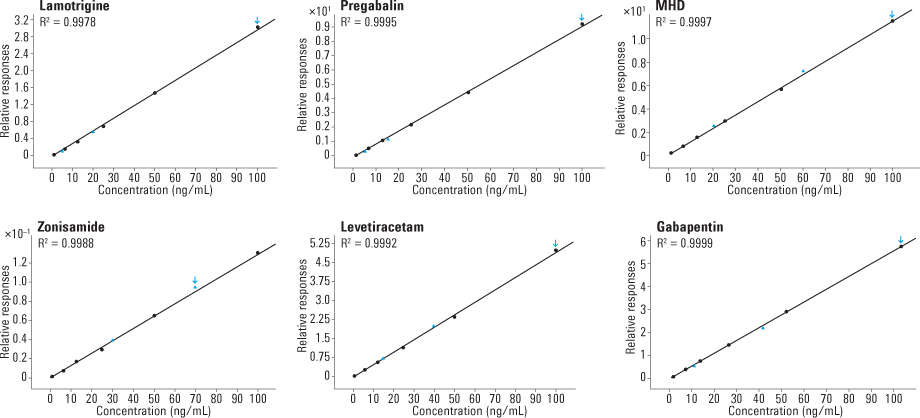
Figure 3. Representative calibration curves show linear range from 1 to 100 mcg/mL for each of the six AED analytes. Dark circles are calibrators and blue triangles are QC standards.
Simple and quick sample preparation
We prepared samples by spiking AEDs into drug-free human serum followed by a protein precipitation with methanol and acetonitrile. We diluted samples 50-fold with water that contained internal standards and then analyzed them (Figure 2) with SPE/MS/MS using the RapidFire/MS/MS system and a reversed-phase C18 cartridge.
High-quality quantitative measurements
To assess carryover, we analyzed a matrix blank immediately after the highest calibrator for all analytes and observed no significant carryover for any of the analytes. Standard curves consisting of each AED spiked into serum demonstrated excellent linearity within the measured range (1–100 mcg/mL), with an R2 value greater than 0.995 (Figure 3).
QC standards for each AED were run over a series of days to establish both intra- and inter-day precision and accuracy. The accuracies were within 7 percent and coefficient of variation values were all less than 7 percent for concentrations within the measured range.
We evaluated the reproducibility of the method by measurement of more than 2,000 sequential injections of all six AEDs spiked into serum. The instrument response was stable for each of the six analytes, with coefficients of variation that ranged from 1.6 to 7 percent. These results show the robustness of the RapidFire system, lifetime of the SPE cartridge, and consistency of quantitation for the analytes in the assay.
Quick, accurate, precise method
This procedure, consisting of a protein precipitation followed by dilute and shoot sample preparation and quick analysis on an Agilent RapidFire/MS/MS, provides a very efficient method to screen and quantitate antiepileptic drugs in human serum. We quickly, accurately, and precisely measured the six drugs in serum using this simple protocol.
The analysis required only 14 seconds per sample, providing a high-throughput method that can analyze more than 250 samples per hour. This methodology delivers comparable results to LC/MS/MS, but at greater than 10-fold the speed and efficiency of typical LC/MS/MS methods.
Greater efficiency and reduced costs
We initially collaborated with a very knowledgeable customer to develop this application. The customer has now validated similar methods and is using RapidFire/MS/MS routinely for clinical research samples. The key benefits of RapidFire/MS to this clinical research lab are:
- Comparable results to traditional LC/MS/MS analyses
- Greater than 10-fold decrease in MS-based analysis time
- Shorter turnaround times for delivery of analytical results
- Annualized laboratory cost savings greater than $200,000 per year
If you need a very efficient method to screen and quantitate AEDs in serum, explore the complete Application Note 5991-2292EN. Then watch this video to learn how the Agilent RapidFire Technology can reduce time and cost for your lead discovery, metabolism, and clinical research applications.
>> Update My Profile | Subscribe to Access Agilent | Article Directory