PD-L1 IHC 28-8 pharmDx for Gastric Cancer


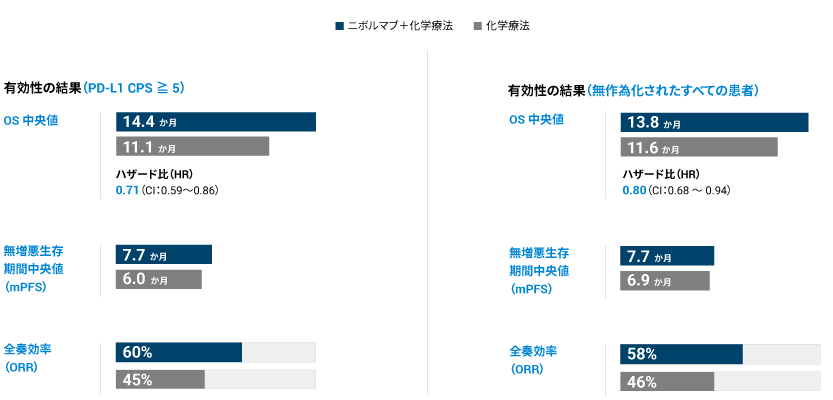
CHECKMATE-649 is a Phase 3, randomized, multi-center, open-label study in patients with previously untreated, non-HER2-positive, advanced or metastatic gastric cancer whose tumors express PD-L1 with a combined positive score (CPS) ≥ 5.
CHECKMATE-649 results highlight overall survival (OS) benefit from OPDIVO® (nivolumab) in combination with fluoropyrimidine and
platinum-containing chemotherapy in non-HER2-positive patients whose tumors express PD-L1 with a combined positive score (CPS) ≥ 5.
PD-L1 IHC 28-8 pharmDx is MHLW-approved and fully validated with analytical performance having met all pre-determined acceptance criteria for sensitivity, specificity and precision.
| Selected analytical validation parameters | Description |
|---|---|
| Specificity |
|
| Sensitivity |
|
| Repeatability |
|
| Reproducibility |
|
OA = Overall Agreement
| Product | Code |
|---|---|
| PD-L1 IHC 28-8 pharmDx | SK005 |
|
Required but not included in kit: Autostainer Link 48 EnVision FLEX Wash Buffer, 20x EnVision FLEX Hematoxylin (Link) PT Link PT Link Rinse Station |
AS480 K8007 K8008 PT101 / PT200 PT109 |
- CHECKMATE-649.
- PD-L1 IHC 28-8 pharmDx Instructions for Use.
- OPDIVO® package insert. Manufactured by: Bristol-Myers Squibb Company, Princeton, NJ 08543 USA U.S. License No. 1713.
Intended Use
For in vitro diagnostic use.
To measure the proportion of PD-L1 expression in cancer tissue or cells
- Indication as an aid for proper administration of nivolumab [recombinant] in non-small cell lung cancer [NSCLC] patients, head-and-neck cancer patients, and gastric cancer patients
- Indication as an aid for proper administration of nivolumab [recombinant] and ipilimumab combination in melanoma patients
It is desirable to measure PD-L1 expression by 28-8 kit in determining whether or not the following drugs can be administered.
- Nivolumab [recombinant] for patients with non-squamous NSCLC or head-and-neck cancer who have been treated with chemotherapy previously
- Combination therapy of nivolumab [recombinant] and chemotherapy for NSCLC patients or gastric cancer patients who have not been treated with chemotherapy
- Combination therapy of nivolumab [recombinant] and ipilimumab for melanoma patients
If it is not possible to measure PD-L1 expression using PD-L1 IHC 28-8 pharmDx, refer to the packaging insert of the drugs, and appropriately judge the adequacy of administration.
See the OPDIVO® product label for specific clinical circumstances guiding PD-L1 testing




