For in vitro diagnostic use
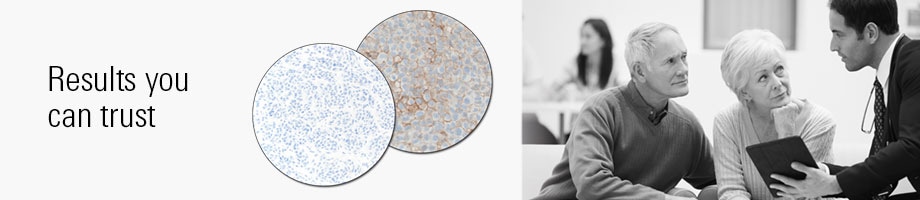
PD-L1 IHC 28-8 pharmDx is a qualitative immunohistochemical assay using Monoclonal Rabbit Anti-PD-L1, Clone 28-8 intended for use in the detection of PD-L1 protein in formalin-fixed, paraffin-embedded (FFPE) non-squamous non-small cell lung cancer (NSCLC), squamous cell carcinoma of the head and neck (SCCHN), and melanoma tissues using EnVision FLEX visualization system on Autostainer Link 48. PD-L1 protein expression is defined as the percentage of evaluable tumor cells exhibiting partial or complete membrane staining at any intensity, as defined by the specific tumor indication staining interpretation guidelines in the instructions for use (IFU).
PD-L1 expression as detected by PD-L1 IHC 28-8 pharmDx in non-squamous NSCLC and SCCHN may be associated with enhanced survival from OPDIVO® (nivolumab) treatment.
PD-L1 expression as detected by PD-L1 IHC 28-8 pharmDx in melanoma may be used as an aid in the assessment of patients for whom OPDIVO® (nivolumab) and YERVOY® (ipilimumab) combination treatment is being considered.
- The only FDA-approved test for PD-L1 expression associated with enhanced survival with OPDIVO for non-squamous NSCLC
- The first and only FDA approved PD-L1 test for use in the assessment of the magnitude of treatment effect on progression free survival in melanoma patients from OPDIVO

- Integrate PD-L1 IHC 28-8 pharmDx without changing staining lab workflow
- Ready-to-use reagents and cell line controls optimized for Autostainer Link 48
- Pre-programmed, validated protocol
Non-squamous NSCLC
Melanoma
SCCHN

Read about the interpretation of PD-L1 IHC 28-8 pharmDx staining results.
Interpretation of non-squamous NSCLC staining results
Interpretation of melanoma staining results
Interpretation of SCCHN staining results
Sign up now for new interactive online PD-L1 interpretation program
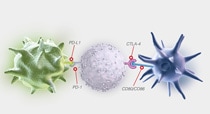
Learn about the CTLA-4 and PD-1/PD-L1 pathways

PD-L1 IHC 28-8 pharmDx is a complete kit with reagents sufficient for 50 tests (50 slides incubated with primary antibody to PD-L1 and 50 slides incubated with the corresponding negative control reagent) and 15 Control Slides for use on Autostainer Link 48.
- EnVision FLEX Target Retrieval Solution, Low pH, 50x
- Peroxidase-Blocking Reagent
- Monoclonal Rabbit Anti-PD-L1, Clone 28-8
- Negative Control Reagent
- PD-L1 IHC 28-8 pharmDx Rabbit LINKER
- Visualization Reagent-HRP
- DAB+ Substrate Buffer
- DAB+ Chromogen
- DAB Enhancer
- Control Slides
| Product | Code |
|---|---|
| PD-L1 IHC 28-8 pharmDx | SK005 |
Required but not included in kit: Autostainer Link 48 EnVision FLEX Wash Buffer, 20x EnVision FLEX Hematoxylin (Link) PT Link |
AS480 K8007 K8008 PT100 |
Contact your local sales representative
Product details, specification sheet and safety data sheet (SDS).