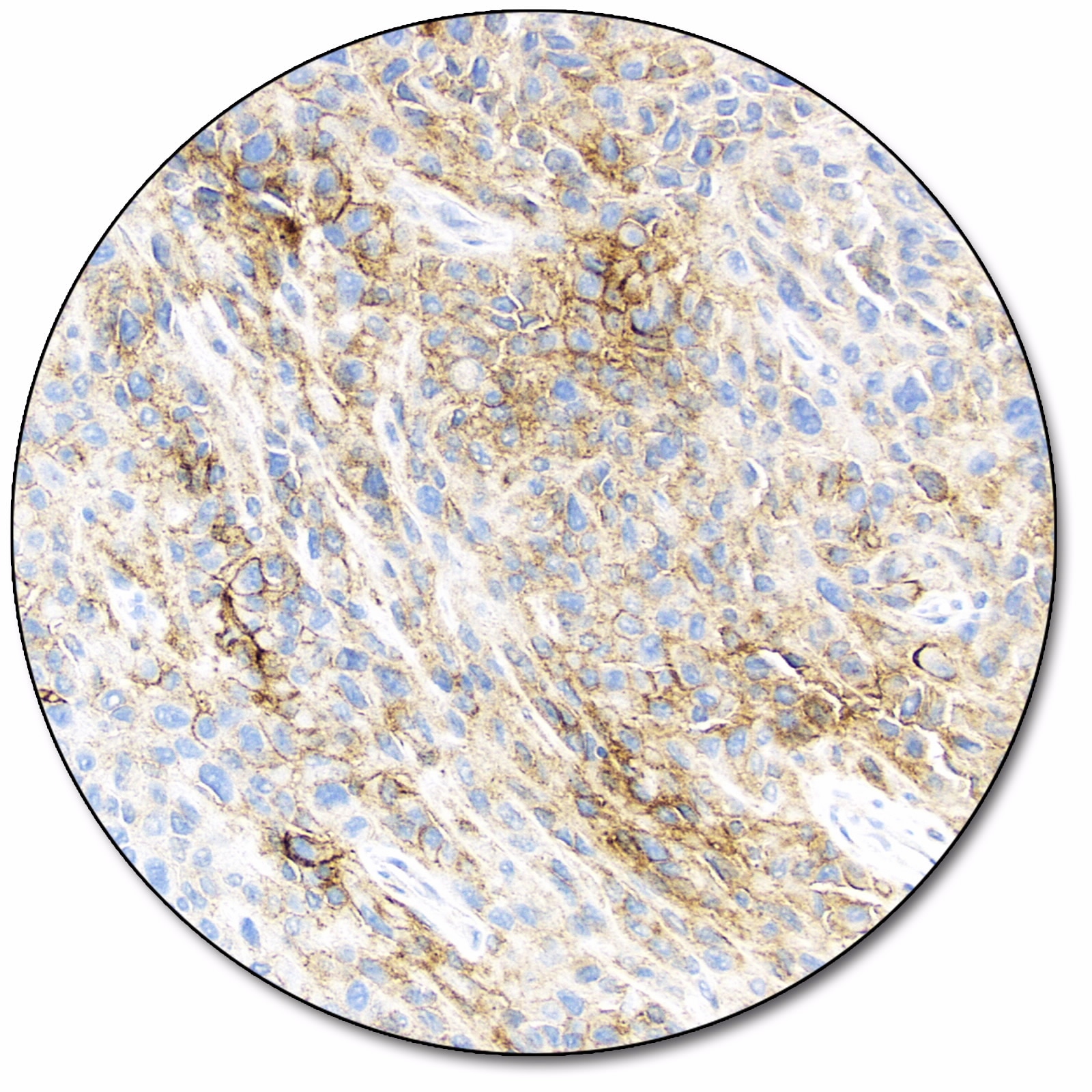
Melanoma
PD-L1 IHC 28-8 pharmDx for Autostainer Link 48
IVD

PD-L1 protein expression in NSCLC, non-squamous non-small cell lung cancer (nsNSCLC), melanoma, MIUC, and esophageal squamous cell carcinoma (ESCC) is determined by using % tumor cell expression, which is the percentage of evaluable tumor cells exhibiting partial or complete membrane staining at any intensity.
Non-small cell lung cancer (NSCLC)
PD-L1 expression (≥ 1% tumor cell expression) as detected by PD-L1 IHC 28-8 pharmDx is indicated as an aid in identifying early stage NSCLC patients for treatment with OPDIVO® (nivolumab) in combination with platinum-doublet chemotherapy as a companion diagnostic test.
Melanoma (OPDUALAGTM)
PD-L1 expression (< 1% tumor cell expression) as detected by PD-L1 IHC 28-8 pharmDx is indicated as an aid in identifying melanoma patients for treatment with Opdualag™ (nivolumab and relatlimab) as a companion diagnostic test.
Non-squamous non-small cell lung cancer (nsNSCLC)
PD-L1 expression (≥ 1% or ≥ 5% or ≥ 10% tumor cell expression) as detected by PD-L1 IHC 28-8 pharmDx in nsNSCLC may be associated with enhanced survival from OPDIVO® (nivolumab).
Melanoma (OPDIVO® and YERVOY®)
PD-L1 expression (≥ 1% or ≥ 5% tumor cell expression) as detected by PD-L1 IHC 28-8 pharmDx in melanoma may be used as an aid in the assessment of patients for whom OPDIVO® (nivolumab) and YERVOY® (ipilimumab) combination treatment is being considered.
Muscle Invasive Urothelial Carcinoma (MIUC)
PD-L1 expression (≥ 1% tumor cell expression) as detected by PD-L1 28-8 pharmDx in MIUC is indicated as an aid in identifying MIUC patients for treatment with OPDIVO® (nivolumab).
Esophageal Squamous Cell Carcinoma (ESCC)
PD-L1 expression (≥ 1% tumor cell expression) as detected by PD-L1 28-8 pharmDx in ESCC indicated as an aid in identifying ESCC patients for treatment with OPDIVO® (nivolumab) in combination with fluoropyrimidine and platinum-based chemotherapy or OPDIVO® (nivolumab) in combination with YERVOY® (ipilimumab).
PD-L1 IHC 28-8 pharmDx is subject to an exclusive trademark license to Agilent Technologies, Inc. OPDIVO®, YERVOY®, and OPDUALAGTM are trademarks owned by Bristol-Myers Squibb.
For more details about PD-L1 IHC 28-8 pharmDx, refer to eIFU
Reg. Status: CE-IVD (IVDR certification)
For In Vitro Diagnostic Use.