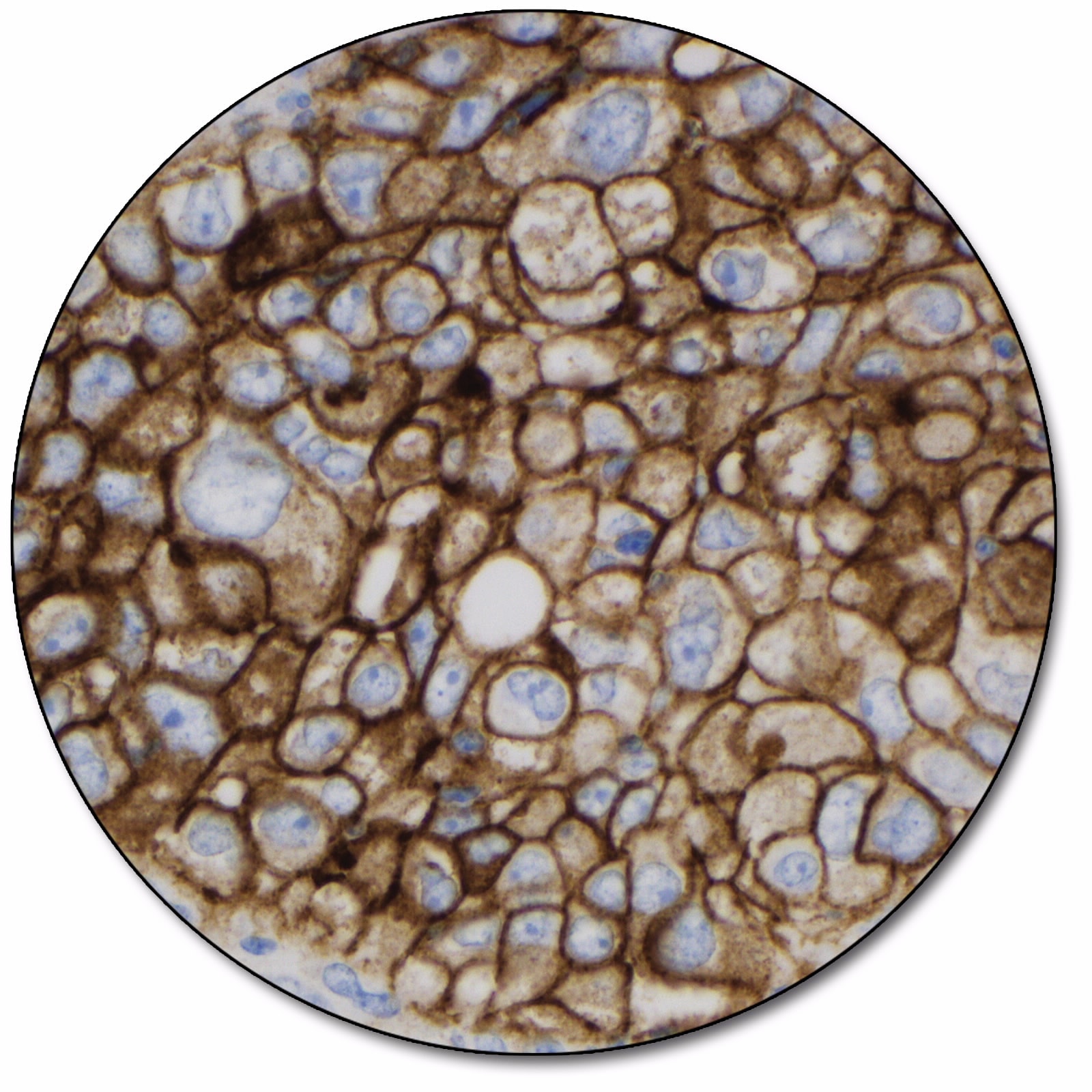
Non-small cell lung cancer
PD-L1 IHC 22C3 pharmDx for Autostainer Link 48
IVD

PD-L1 IHC 22C3 pharmDx is a qualitative immunohistochemical assay using Monoclonal Mouse Anti-PD-L1, Clone 22C3 intended for use in the detection of PD-L1 protein in formalin-fixed, paraffin-embedded (FFPE) non-small cell lung cancer (NSCLC), urothelial carcinoma, and head and neck squamous cell carcinoma (HNSCC) tissues using EnVision FLEX visualization system on Autostainer Link 48.
Non-Small Cell Lung Cancer (NSCLC)
PD-L1 protein expression in NSCLC is determined by using Tumor Proportion Score (TPS), which is the percentage of viable tumor cells showing partial or complete membrane staining at any intensity. The specimen should be considered to have PD-L1 expression if TPS ≥ 1%.
PD-L1 IHC 22C3 pharmDx is indicated as an aid in identifying NSCLC patients for treatment with KEYTRUDA® (pembrolizumab). See the KEYTRUDA® product label for expression cutoff values guiding therapy in specific clinical circumstances.
Urothelial Carcinoma
PD-L1 protein expression in urothelial carcinoma is determined by using Combined Positive Score (CPS), which is the number of PD-L1 staining cells (tumor cells, lymphocytes, macrophages) divided by the total number of viable tumor cells, multiplied by 100. The specimen should be considered to have PD-L1 expression if CPS ≥ 10.
PD-L1 IHC 22C3 pharmDx is indicated as an aid in identifying urothelial carcinoma patients for treatment with KEYTRUDA® (pembrolizumab). See the KEYTRUDA® product label for specific clinical circumstances guiding PD-L1 testing.
Head and Neck Squamous Cell Carcinoma (HNSCC)
PD-L1 protein expression in HNSCC is determined by using Combined Positive Score (CPS), which is the number of PD-L1 staining cells (tumor cells, lymphocytes, macrophages) divided by the total number of viable tumor cells, multiplied by 100. The specimen should be considered to have PD-L1 expression if CPS ≥ 1.
PD-L1 IHC 22C3 pharmDx is indicated as an aid in identifying HNSCC patients for treatment with KEYTRUDA® (pembrolizumab). See the KEYTRUDA® product label for specific clinical circumstances guiding PD-L1 testing.
PD-L1 IHC 22C3 pharmDx
The kit includes reagents required for the immunohistochemical staining (except wash buffer), control slides representing different expression levels of PD-L1 protein, and detailed instructions. The kit has been tailored especially for use on Autostainer Link 48 instruments. The materials provided are sufficient for 50 tests (50 slides incubated with monoclonal mouse antibody to PD-L1 and 50 slides incubated with the corresponding negative control reagent, 100 slides in total).
KEYTRUDA® is a registered trademark of Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA
For In Vitro Diagnostic Use.
