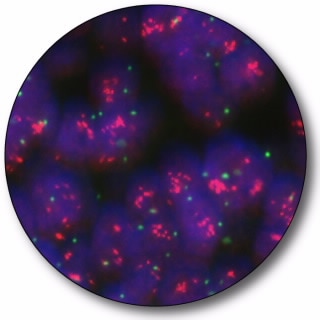
HER2 IQFISH pharmDx

HER2 IQFISH pharmDx is designed to quantitatively determine HER2 gene amplification in formalin-fixed, paraffin-embedded (FFPE) breast cancer tissue specimens and FFPE specimens from patients with metastatic gastric or gastroesophageal junction adenocarcinoma. Gene amplification is determined from the ratio between the number of signals from the hybridization of the HER2 gene probe (red signals) and the number of signals from the hybridization of the CEN-17 reference chromosome 17 probe (green signals).
HER2 IQFISH pharmDx is indicated as an aid in the assessment of breast and gastric patients for whom Herceptin® treatment is being considered and for breast cancer patients for whom PERJETA™ or KADCYLA™ treatment is being considered (see Herceptin®, PERJETA™, and KADCYLA™ package inserts). For breast cancer patient, results from the HER2 IQFISH pharmDx are intended for use as an adjunct to the information currently used for estimating prognosis in stage II, node-positive breast cancer patients.
HER2 IQFISH pharmDx is a complete system providing all reagents required to perform 20 assays. A standard validated procedure and validated interpretation guidelines are also provided.
HercepTest, Herceptin®, PERJETA™ and KADCYLA™ are trademarks owned by Genentech, Inc. and/or F. Hoffmann-La Roche Ltd.; HercepTest is subject to an exclusive trademark license to Dako Denmark A/S.