Access Agilent eNewsletter October 2016

Efficient analytical methods for catecholamines, metanephrines, and related metabolites in urine and plasma
Linda Côté
Agilent Clinical LC/MS Application Specialist
Christophe Deckers
Agilent Sample Preparation Specialist
In recent years, modern sample preparation techniques coupled with more sensitive LC/MS/MS systems have advanced clinical research methodology. With these advances, many mid-sized clinical research labs are required to perform multiple types of analyses with only one LC/MS/MS system. To enable overnight analyses, these labs must develop methods that use the same LC column and mobile phase on this single system. To achieve this goal, we developed a unified method that collapsed seven LC/MS/MS methods into only four. The key to this unified method was the use of well-chosen, selective sample preparation products.
Determinations of catecholamines, metanephrines, and related metabolites—such as vanillylmandelic acid (VMA), homovanillic acid (HVA), and 5-hydroxyindoleacetic acid (5-HIAA)—in various matrices have been done for many years. Classical methods use long and proprietary sample preparation procedures with long chromatography methods that employ ion-pairing reagents and electrochemical detectors. Due to lack of detector specificity, these methods require run times as long as 30 minutes to separate possible matrix interferences.
Seven methods reduced to only four
In the past, seven methods were necessary to analyze catecholamines, metanephrines, and related metabolites in matrices such as urine and plasma at analyte concentrations that range from
- µg/mL to ng/mL for urine
- pg/mL for plasma
Looking at ways to streamline the process, we developed a unified approach where the same Agilent Pursuit PFP column and the same Agilent 1290 Infinity II LC System and Agilent 6460 Triple Quadrupole LC/MS were used to analyze multiple compounds and matrices. This unification was achieved with novel and selective sample preparation techniques that were adapted to the matrix and to the desired lower limit of quantification (LLOQ). As a result, we reduced the seven methods to four analytical methods that are robust, sensitive, and precise:
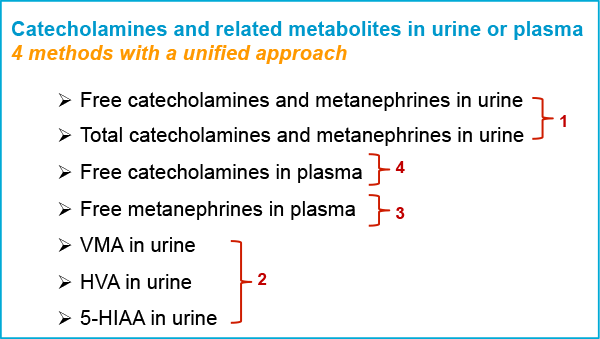
Figure 1. Increased productivity: By combining methods, we reduced seven analyses to only four, while maintaining excellent analytical results.
- Urinary catecholamines, metanephrines, and 3-methoxytyramine in a single LC/MS/MS run using Agilent Bond Elut Plexa SPE
- Urinary vanillylmandelic, homovanillic, and 5-hydroxyindoleacetic acids by LC/MS/MS after sample dilution
- Plasma metanephrines and 3-methoxytyramine by LC/MS/MS using Agilent SampliQ WCX SPE
- Plasma catecholamines by LC/MS/MS using Agilent Captiva NDLipids and Agilent Bond Elut PBA SPE
Figure 1 shows how we combined analyses. Red numbers in the figure provide the order in which the methods were combined.
Methods successfully combined
Table 1 summarizes the combined methods. It shows that the unified method achieved the necessary linear ranges (orange) with a single chromatography column and LC/MS/MS system (dark green). The mobile phase (blue) was the same for three of the four analyses, which made it easy to expand productivity with overnight runs. Optimized sample preparation was crucial for successful method integration. Another contributor to success was the Agilent Pursuit PFP column, which uniquely retains these very hydrophilic compounds without the use of ion pairing modifiers. Figure 2 shows the excellent chromatography for catecholamines and metanephrines in a single six-minute run.
| 1. Free and total catecholamines and metanephrines | 2. VMA, HVA, 5-HIAA | 3. Metanephrines | 4. Catecholamines | |
|---|---|---|---|---|
| Matrix | Urine | Urine | Plasma | Plasma |
| Linear range | 1.56-3000 ng/mL | 0.08-100 mg/L | 15.6-10000 pg/mL | 5-2500 pg/mL |
| Sample prep | Boronate complex/ Bond Elut Plexa SPE | Dilution | WCX SPE | Captiva NDLipids/BondElut PBA SPE |
| Chromatography | Pursuit-PFP | Pursuit-PFP | Pursuit-PFP | Pursuit-PFP |
| Mobile phase | 0.2% formic acid—methanol | 0.2% formic acid—methanol | 0.2% formic acid—methanol | 1 mM ammonium fluoride—methanol |
| LC/MS/MS | 1290/6460 | 1290/6460 | 1290/6460 | 1290/6460 |
Table 1. Unified approach in method development resulted in four methods that shared the same Agilent chromatography column and LC/MS/MS system.
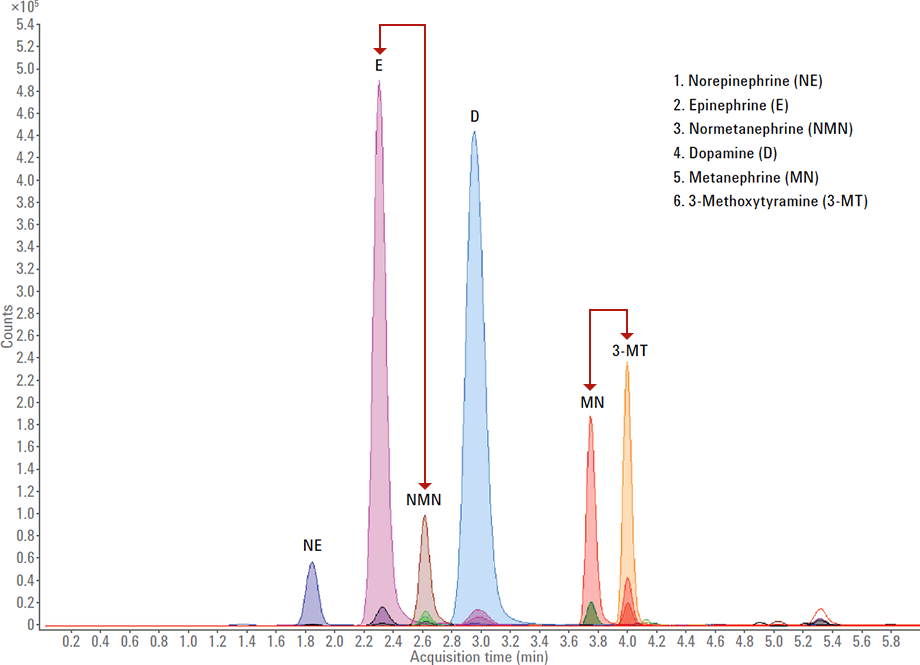
Figure 2. Baseline resolution for catecholamines and metanephrines in a single run. Arrows highlight separation of critical peak pairs.
Dependable quantitative results from optimized sample preparation
Unique sample preparation techniques gave us the necessary limits of quantification (LOQs), linearity, accuracy, and precision that each method required.
Method 4 in Table 1 was the most challenging to implement for two reasons. First, catecholamines are readily oxidized in plasma. To maintain sample stability for this analysis, you must keep plasma frozen at -80 °C and then add ETDA and sodium metabisulfite stabilizer solutions immediately before analysis. Second, to measure low pg/mL analytes in plasma, you need a two-step sample cleanup. We tried solid phase extraction (SPE) alone and despite aggressive wash steps, the samples still needed additional cleanup.
In our final method, we first accomplished a protein precipitation in a special lipid-removal filtration cartridge – Agilent Captiva NDLipids. We then took that filtrate, depleted of proteins and lipids, and proceeded to a highly selective solid phase extraction (SPE) cartridge – Agilent Bond Elut PBA SPE. That SPE phase has phenyl boronic acid (PBA) functional groups, which are highly selective towards the cis-diols in catecholamines.
Finally, to obtain the desired LLOQ for this method, we used a mobile phase of freshly prepared 1 mM ammonium fluoride in water. With these optimizations, we achieved acceptable accuracy and precision even at very low pg/mL concentrations.
Improved laboratory productivity from streamlined, unified approach
In summary, we developed a unified approach where the same chromatography principles and the same modern LC/MS/MS instrumentation were used to analyze multiple compounds and matrices. By doing so, we adapted novel and selective sample preparation techniques to the matrix and to the desired LLOQ. We reduced seven analytical methods to four methods that were robust, sensitive, and precise.
For additional information, view an exciting webinar that provides all the details about this research. Then learn more about Agilent sample preparation solutions and triple-quadrupole LC/MS instrumentation.
For Research Use Only. Not for use in diagnostic procedures.
Stay informed about the applications that are important to you
Subscribe to Access Agilent
Our free customized
monthly eNewsletter
Article Directory – October 2016
All articles in this issue
 Designed for true mobility—the 490-Mobile takes your lab to the sample
Designed for true mobility—the 490-Mobile takes your lab to the sample Agilent 1260 Infinity II solution delivers fast, sensitive, and automated amino acid analysis
Agilent 1260 Infinity II solution delivers fast, sensitive, and automated amino acid analysis Efficient analytical methods for catecholamines, metanephrines, and related metabolites in urine and plasma
Efficient analytical methods for catecholamines, metanephrines, and related metabolites in urine and plasma Agilent IDP-3 Dry Scroll Pump provides clean, quiet, and reliable GC/MSD analysis
Agilent IDP-3 Dry Scroll Pump provides clean, quiet, and reliable GC/MSD analysis Determination of chemical species in marine fuel oil by multidimensional GC/MS analysis
Determination of chemical species in marine fuel oil by multidimensional GC/MS analysis Tip: Easily transfer Agilent GC/MSD methods to Agilent triple quadrupole GC/MS systems
Tip: Easily transfer Agilent GC/MSD methods to Agilent triple quadrupole GC/MS systems Agilent ICP-QQQ provides unique solutions for nuclear applications
Agilent ICP-QQQ provides unique solutions for nuclear applications
Figure 1

Increased productivity: By combining methods, we reduced seven analyses to only four, while maintaining excellent analytical results.
Figure 2

Baseline resolution for catecholamines and metanephrines in a single run. Arrows highlight separation of critical peak pairs.