Access Agilent eNewsletter March 2016

Multiple heart-cutting 2D-LC/MS method for reproducible resolution of chiral drug metabolites
Siji Joseph, Agilent Application Scientist
The analysis of drug metabolites in complex biological matrices is analytically challenging because of their low abundance. Additionally, metabolites of interest are often isobaric and have similar fragmentation patterns, so unambiguous identification is difficult with conventional LC/MS analysis. Chiral drugs and their metabolites provide additional challenges. For drug metabolism and pharmacokinetics (DMPK) studies and regulatory requirements, it is important to obtain chiral resolution of metabolites and have reproducible methods in place to monitor them. The Agilent 1290 Infinity II 2D-LC Solution with multiple heart-cutting provides excellent results in this demanding situation.
Difficult separation problem
Warfarin—a widely used racemic anticoagulant, primarily undergoes oxidative metabolism to give 4’-, 6-, 7-, 8-, and 10-hydroxylated metabolites. Because these hydroxylated metabolites are isobaric and have similar fragmentation patterns, completely resolved chromatographic peaks are important for confident measurement of trace-level metabolites with mass spectrometry.
The use of supercritical fluid chromatography (SFC) coupled with triple quadrupole (QQQ) mass spectrometry for simultaneous identification and quantitation of warfarin and hydroxylated metabolites was presented previously in Agilent publication 5991-5725EN. A multiple heart-cutting 2D-LC/quadrupole-time-of-flight (Q-TOF) methodology for the successful achiral and chiral separation of warfarin and five major hydroxylated metabolites is described here.
Multiple heart-cutting enables resolution of all peaks
This study was performed using an Agilent 1290 Infinity II 2D-LC Solution coupled with an Agilent 6530 Accurate-Mass Quadrupole Time-of-Flight (Q-TOF) LC/MS system. The 2D LC/MS method employed an achiral reversed-phase separation in the first LC dimension (phenyl hexyl stationary phase) followed by transfer of warfarin and hydroxywarfarin metabolites through multiple heart-cuts to the second dimension chiral column (Chiral OD-3R and/or Chiral AD-3R). This method enabled resolution of the chiral drug and its metabolites prior to high-resolution accurate-mass MS and MS/MS analysis.
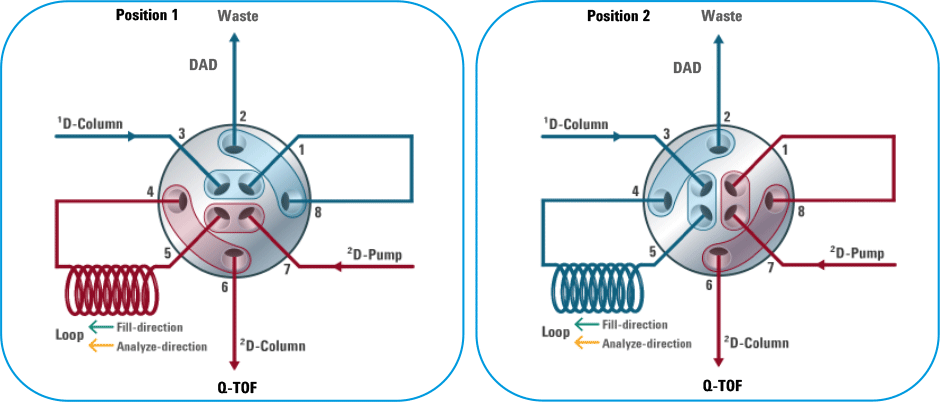
Figure 1. Schematic diagram of the 2-position/4-port-duo valve for precise heart-cutting applications.
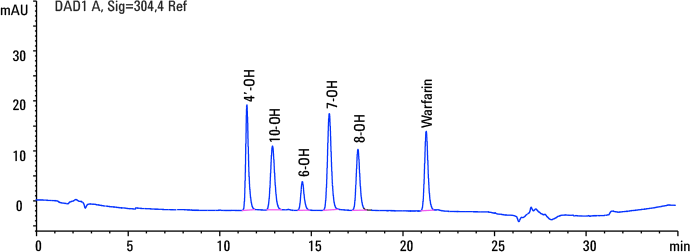
Figure 2. UV data shows outstanding first-dimension achiral separation with the Agilent ZORBAX Eclipse Plus Phenyl Hexyl column.
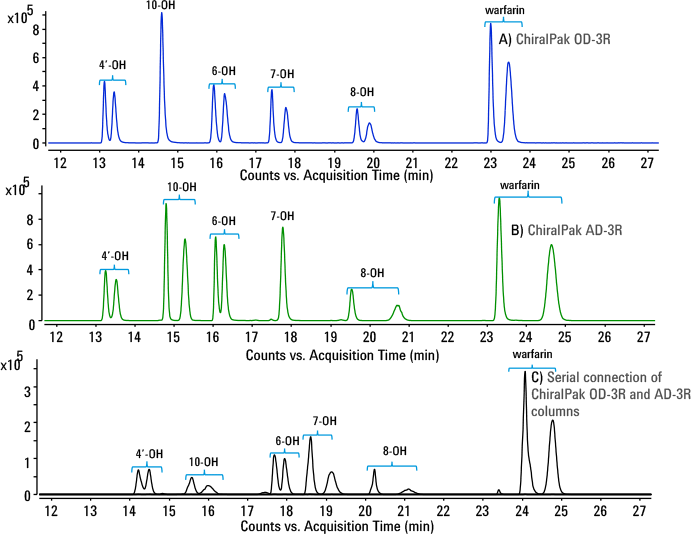
Figure 3. Stereoselective separation of racemic pairs of hydroxywarfarins as well as parent R/S-warfarin in the second dimension using ChiralPak columns.

Figure 4. A 6-position/14-port valve with cluster of six pre-installed sampling loops simplifies storage of six heart-cuts.
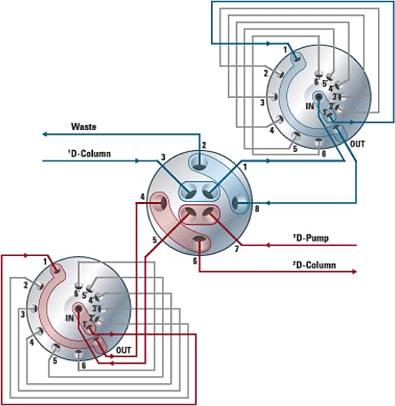
Figure 5. Smart valve-loop setup can store eluents from 12 heart-cuts. It incorporates a 2-position/4-port duo-valve (shown in center) plus two 6-position/14-port valves.
A diode array detector (DAD) was used to record the UV signal after the first LC dimension and to determine the heart-cut timings. A 2-position/4-port-duo valve was used to perform the multiple heart-cuts (Figure 1). When the valve was in Position 1, the first dimension column eluent was directed to the DAD and then to waste. When the valve was in Position 2, heart-cutting took place. The first-dimension column eluent was directed into an 80-µL loop. The loop was filled for a predetermined duration of 0.6 min, based on peak width of the desired analyte. A similar strategy was used to perform hear-cutting for other peaks. Agilent LC/MS ChemStation software (C01.03 with 2D-LC add-on) enabled easy and precise control of 2D-LC heart-cutting.
Stereospecific resolution of drug metabolites
The Agilent ZORBAX Eclipse Plus Phenyl Hexyl reversed-phase column showed excellent baseline separation of warfarin and all five hydroxylated metabolites with a resolution greater than 4 (Figure 2).
Use of the ChiralPak OD-3R column in the second dimension enabled baseline separation of enantiomeric pairs of four of the five regio-isomers of hydroxywarfarin, but it did not separate the 10-hydroxywarfarin enantiomers (Figure 3A). Use of the ChiralPak AD-3R column separated enantiomeric pairs of four of the five regio-isomers of hydroxywarfarin, including the 10-hydroxywarfarin, but did not separate the 7-hydroxywarfarin enantiomers (Figure 3B). However, a serial combination of ChiralPak OD-3R and AD-3R columns in the second LC dimension was able to resolve enantiomers of all five hydroxywarfarin regio-isomers, as well as parent warfarin—a total of twelve peaks in less than 25 minutes (Figure 3C). The high pressure limit of the Agilent 1290 Infinity II LC supported coupling of two columns with different column chemistries and offered superior separation of metabolite enantiomers.
Hardware advancements deliver successful multiple heart-cutting analysis
Multiple heart-cutting is much easier with the new, improved Agilent multiple heart-cutting valve kit. A 6-position/14-port valve is included in the multiple heart-cutting 2D-LC upgrade kit (G4242A) and can be connected to the 2-position/4-port duo-valve (5067-4170). The 6-position/14-port valve has a cluster of six pre-installed sampling loops and can easily store six heart-cut eluents from the first-dimension column (Figure 4).
If you need more heart-cuts, the duo-valve can be coupled with two 2-position/4-port duo-valves. This valve configuration provides two parking decks with 12 loop positions (Figure 5). The new software “multiple heart-cutting viewer” enables you to perform and monitor multiple heart-cutting experiments with ease.
Preconfigured hardware simplifies a difficult analysis
To accurately analyze low-abundance isobaric chiral drugs and their metabolites, you need to understand the inherent challenges. Given their similar mass spectral fragmentation patterns, complete chromatographic separation is a must. As illustrated by this successful separation of warfarin and enantiomeric pairs of five major hydroxylated metabolites, you can confidently use multiple heart-cutting 2D-LC in combination with Q-TOF detection to accomplish this analysis with ease.
Discover today how the Agilent 1290 Infinity II 2D-LC Solution makes it easier for your lab to use 2D-LC to solve difficult separation problems.
Stay informed about the applications that are important to you
Subscribe to Access Agilent
Our free customized
monthly eNewsletter
Article Directory – March 2016
All articles in this issue
 Evolution of multiple heart-cutting 2D-LC: Agilent 1290 Infinity II provides high-resolution sampling and easy quantitation
Evolution of multiple heart-cutting 2D-LC: Agilent 1290 Infinity II provides high-resolution sampling and easy quantitation Tip: How to achieve efficient, effortless LC method transfer to the Agilent 1290 Infinity II LC System
Tip: How to achieve efficient, effortless LC method transfer to the Agilent 1290 Infinity II LC System Agilent 1290 Infinity II RID delivers high resolution and rapid polymer characterization
Agilent 1290 Infinity II RID delivers high resolution and rapid polymer characterization Dual-needle option for LC autosamplers provides improved speed, flexibility, and accuracy
Dual-needle option for LC autosamplers provides improved speed, flexibility, and accuracy Multiple heart-cutting 2D-LC/MS method for reproducible resolution of chiral drug metabolites
Multiple heart-cutting 2D-LC/MS method for reproducible resolution of chiral drug metabolites Quick, accurate screening for suspect environmental contaminants with Agilent solutions
Quick, accurate screening for suspect environmental contaminants with Agilent solutions
Figure 1

Schematic diagram of the 2-position/4-port-duo valve for precise heart-cutting applications.
Figure 2

UV data shows outstanding first-dimension achiral separation with the Agilent ZORBAX Eclipse Plus Phenyl Hexyl column.
Figure 3

Stereoselective separation of racemic pairs of hydroxywarfarins as well as parent R/S-warfarin in the second dimension using ChiralPak columns.
Figure 4

A 6-position/14-port valve with cluster of six pre-installed sampling loops simplifies storage of six heart-cuts.
Figure 5

Smart valve-loop setup can store eluents from 12 heart-cuts. It incorporates a 2-position/4-port duo-valve (shown in center) plus two 6-position/14-port valves.