Access Agilent eNewsletter May 2015
>> Update My Profile | Subscribe to Access Agilent | Article Directory

New Agilent atomic spectroscopy standards ensure the highest levels of accuracy and quality
By Melanie Rothermich
Agilent Product Manager, Chemical Standards
Alejandro Amorin
Agilent Technical Marketing Engineer
Laboratories that wish to establish and maintain a quality management system undertake a daunting and time-consuming task. ISO/IEC 17025 is the global quality standard for the management and technical requirements for testing laboratories. Manufacturers of standards that are accredited to ISO/IEC 17025 must demonstrate that they are technically competent, use documented and validated methods, and are able to produce precise and accurate test and/or calibration data with known uncertainties. Agilent certified inorganic standards meet all these demanding requirements.
Because all sample measurements are made with reference to the initial calibration, the accuracy of the analysis is dependent on the accuracy of the calibration standards. These standards must be free of contaminants, and most importantly, must be certified using the most rigorous and robust techniques. Using Certified Reference Materials (CRMs) for preparation of calibration standards improves accuracy, establishes traceability, and allows quantification of the measurement of uncertainty.
Agilent now offers a complete line of spectroscopy CRMs, manufactured in an ISO 9001, ISO Guide 34 facility and certified in an ISO/IEC 17025 testing laboratory. These CRMs are available for methods that use:
- Atomic absorption spectroscopy (AAS)
- Microwave plasma atomic emission spectroscopy (MP-AES)
- Inductively coupled plasma optical emission spectroscopy (ICP-OES)
- Inductively coupled plasma mass spectrometry (ICP-MS)
With Agilent CRMs, your laboratory is assured of quality, purity, and consistency.
 Enlarge
Enlarge
Figure 1. Certificate of analysis for an Agilent spectroscopy CRM depicts the additional levels of quality control that are applied during the manufacture of the standard.
Agilent CRMs use NIST methods for accuracy and traceability
All Agilent spectroscopy CRMs are certified using the high-performance spectroscopy protocol [1] developed by the National Institute of Standards and Technology (NIST). Both the certified concentrations and uncertainty values are traceable to NIST Standard Reference Materials (SRMs). NIST uses high-performance ICP-OES to certify its SRM 3100 series of spectrometric standards. NIST recommends that other manufacturers of standards use this technique to certify single-element standards with high accuracy, low uncertainty, and direct traceability to the NIST SRM 3100 series.
Figure 1 shows a typical Certificate of Analysis (CoA) for an Agilent spectroscopy CRM. This CoA highlights some of the features of our CRMs that improve quality and productivity. The certified concentrations are reported on the CoA as both weight/weight (µg/g) and weight/volume (µg/mL). The CoA details the method used for certification, intended use, instructions for proper use and recommendations on appropriate conditions of storage.
Trace impurities are assayed using an Agilent ICP-MS and reported on the CoA for ICP-OES/ICP-MS standards. (Trace impurities are not reported for the atomic absorption standards because AA is a single-element technique). The standards have a long shelf life – up to 18 months – supported by long-term stability studies performed as part of the requirements for Guide 34 accreditation.
Agilent AAS, ICP-OES, and ICP-MS standards are made from high-purity raw materials, high-purity acids, and 18-MΩ de-ionized water. They are packed in precleaned, high-purity, high-density polyethylene (HDPE) bottles before they are shipped in poly-sealed bags with tamper-evident seals.
 Enlarge
Enlarge
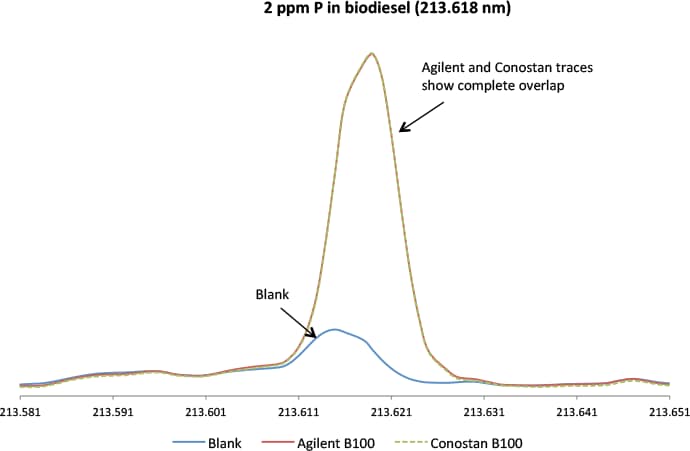
Figure 2. Comparison of Agilent and Conostan Biodiesel B100 standards for phosphorus at 2 ppm (213.618 nm) demonstrates excellent agreement.
 Enlarge
Enlarge
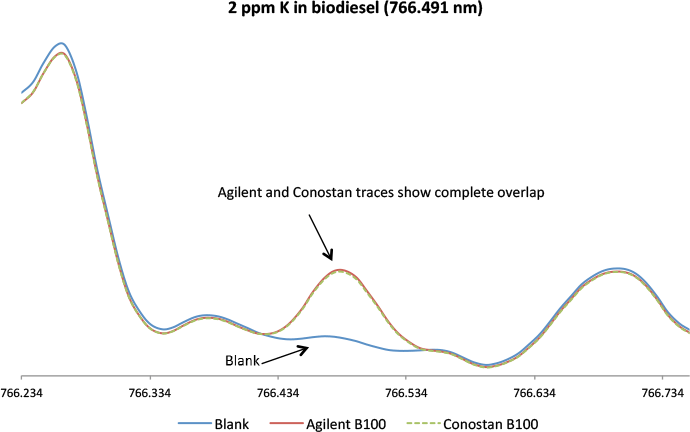
Figure 3. Comparison of Agilent and Conostan Biodiesel B100 standards for potassium at 2 ppm (766.491 nm) shows nearly identical traces.
Agilent standards provide unsurpassed results
Agilent also offers a full range of single and multi-element metallo-organic and biodiesel standards, base oil and pure solvent for the preparation of working standards for analysis of lubricant additives, wear metals and petroleum products. We demonstrated the quality of Agilent standards by comparison with industry-leading Conostan metallo-organic multi-element oil and biodiesel standards. The signal graphics in Figures 2 and 3 reveal an excellent match between the biodiesel standards from Agilent and Conostan. We analyzed phosphorus and potassium because the industry regards these elements as more difficult to measure in biodiesel.
The signal graphics for the Agilent and Conostan standards are virtually identical for the nominated elements. In addition, there is no significant change in the emission spectrum on either side of the analytical wavelength. This baseline indicates that the standard is free of unwanted components that would increase the background. The traces in Figures 2 and 3 confirm that the Agilent standard is “clean” and equivalent to the Conostan standard.
No need to compromise on standards
Agilent quality systems deliver the strictest quality control of incoming raw materials and meticulous validation of standard concentrations, homogeneity, stability, and reporting. So if you need certified reference materials for compliance with ISO 17025 requirements, Agilent CRMs help you achieve precise and accurate calibration data with consistent performance. Explore the entire family and see for yourself.
Need more information about ISO 17025 requirements? Dr Ludwig Huber wrote an excellent introduction to ISO/IEC 17025 as the global standard for laboratory accreditation. His primer, Understanding and Implementing ISO/IEC 17025, is freely available from Agilent (publication number 5990-4540EN).
>> Update My Profile | Subscribe to Access Agilent | Article Directory
Figure 1.
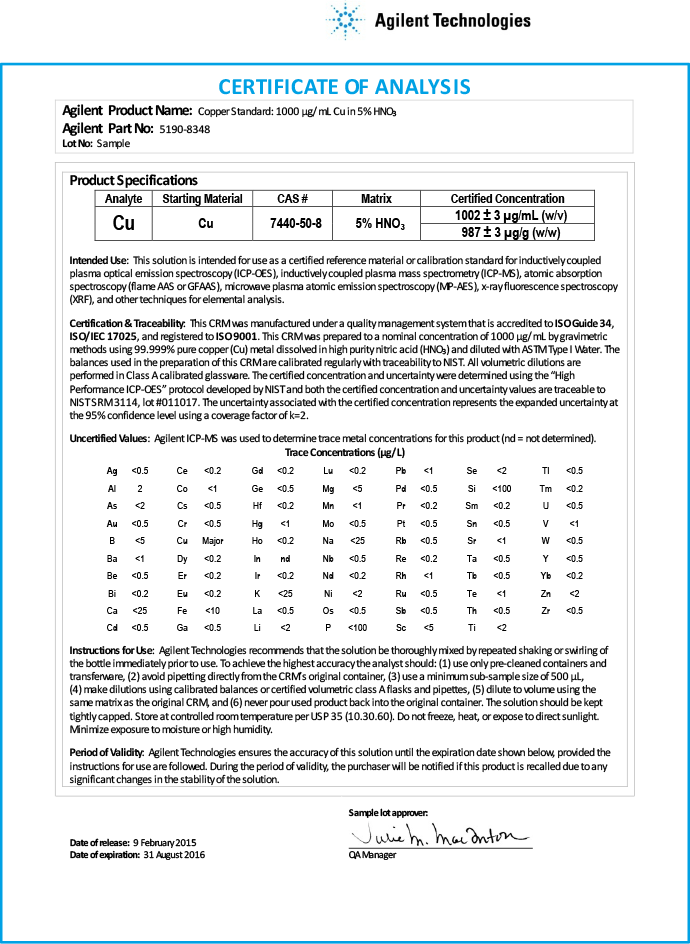
Certificate of analysis for an Agilent spectroscopy CRM depicts the additional levels of quality control that are applied during the manufacture of the standard.