Access Agilent eNewsletter August 2015
>> Update My Profile | Subscribe to Access Agilent | Article Directory

Ask the Expert: How can Agilent solutions help with my lipidomics research?
By Dr. Seetaram Gundimeda
Agilent Application Scientist
Lipidomics – the study of lipids – is a relatively new research area that includes:
- Qualitative and quantitative profiling
- Sample preparation to profile either a particular class of lipids or global lipids
- Identification methods using various detectors
- Biological evaluations that include biosynthesis and activity assays
While lipidomics is a complex area of investigation, a wide range of Agilent tools can accelerate your research on the role that lipids play in biological processes.
 Enlarge
Enlarge
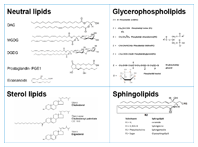
Figure 1. Examples of lipid classes.
Lipids: a complex web of related chemical structures
Lipids are a group of fatty acids and their derivatives. Most of the fatty acid derivatives are based on glycerol, which has three hydroxyl groups. All these hydroxyl groups can be esterified with long chain fatty acids. If one hydroxyl group is esterified it is called a mono acyl glyceride. If two hydroxyl groups are esterified it leads to diglyceride and esterification of the three hydroxyl groups leads to triglycerides. In all the three glycerol based lipids, the fatty acyl chain length can vary from C12 to C24 with varying degrees of unsaturation and hydroxylation. Many times, the third hydroxyl group is linked to a hydrophilic moiety like a sugar, phosphate, phosphoethanolamine, phosphocholine, phosphoserine, or phosphoinositol. This linkage makes a new lipid class – glycerophospholipids (Figure 1).
Sphingolipids are another class where the amine group of a sphingoid backbone makes an amide bond with a free fatty acid (Figure 1). R2 can have different chain lengths with unsaturation at various positions. If R1 has only one sugar, the lipid is called a cerebroside. If the sugar moiety has a sulphate group, then the lipid is called as sulfatide. If R1 has more than one sugar, then this lipid is called a globoside. Here, the sugars are usually a combination of N-acetylgalactosamine, D-glucose, or D-galactose. If R1 has one or more than one sialic acid group (N-acetylneuraminic acid unit), then these molecules are called gangliosides.
Lipids contribute to a vast array of biological processes
Lipids are building blocks of major structural elements of biological membranes. They play crucial roles as enzyme cofactors, intracellular messengers, antigens, electron carriers, chaperones (to help membrane proteins fold), and hormones. Their function in reproductive system and immune system-causing inflammation, fever and pain is discussed widely. Because of their role in cell proliferation, lipids contribute to cancer pathways. Many lipids are involved in metabolic disorders, playing a major role in the progress of Alzheimer’s and Parkinson’s diseases. They occupy a major space in the human metabolome, so we must understand the role of lipids to understand the metabolome comprehensively.
Successful approaches in lipidomics
Lipidomic studies can use two different analysis methods. One approach is based on liquid chromatography/mass spectrometry (LC/MS/MS) and another one is a “shotgun” approach. In the first case, you load the sample onto an LC column, where the constituents are separated into individual peaks and subsequently sent to the MS. In a shotgun approach, you infuse the sample into the MS without separating the components with LC. This latter technique reduces the analysis time drastically and you can improve detection limits for some compounds.
However, the shotgun strategy has its own challenges. When lipids are present in isomeric form, you need MS/MS to correctly identify the isomer. In this approach, MS/MS can give ambiguous results. Some highly ionizing lipids like phosphatidylcholines can suppress the ionization of low ionizing molecules like ether lipids. With LC/MS, you can baseline-separate the compounds, which minimizes the ionization suppression and produces MS/MS spectra that are free from interferences.
Two-dimensional LC/MS delivers more thorough analysis
A lipid mixture extracted from plasma, serum, or tissue contains a variety of lipids with varying degrees of polarity. A normal-phase LC column can separate the complex mixture into individual classes of lipids, but provides less separation among the members of each class. So you typically collect the entire class as a fraction onto a reversed-phase LC column for further separation. This two-dimensional LC gives a more complete analysis of the compounds present in the mixture.
In another approach, you replace the normal-phase LC column with a solid-phase extraction cartridge. Here, you load a lipid mixture onto an aminopropyl cartridge and selectively elute each class with a suitable combination of solvents. If the objective is not exhaustive coverage but rather a moderately broader coverage in a single analysis, then you can use a single reversed-phase LC/MS method. But the analysis must include both positive and negative ionization modes. The majority of lipids ionize very well in negative mode, but for ceramides, positive mode is ideal.
An ideal LC/MS/MS instrument for lipidomic study
Agilent Triple quadrupole LC/MS instruments are ideal for quantitative lipidomics. Ion traps are known to be the most sensitive, but they lack the accurate mass measurement you need to distinguish the closely related lipid masses. For example, monoacyl C17-linked phosphatidylcholine (PC(17:0/0:0)) has an exact mass of 510.3554 and an ether-linked phosphatidylcholine (PC(O-18:0/0:0)) has an exact mass of 510.3918. To detect these two molecules separately, the mass spectrometer must have a resolution of 14,000. You easily can achieve this resolution with Agilent Q-TOF instruments. Lipids show diagnostic ions in their MS/MS spectra. Often, these ions fall in the one-third mass cutoff region in ion trap and can be missed. But a Q-TOF instrument can detect them readily.
The complex field of lipidomics demands a comprehensive set of powerful analytical tools. To discover additional information about our wide-ranging solutions to advance your study of lipids, learn more today about our sample preparation supplies, LC/MS systems, and advanced informatics software.
>> Update My Profile | Subscribe to Access Agilent | Article Directory