Access Agilent eNewsletter, May 2014
>> Update My Profile | Subscribe to Access Agilent | Article Directory

Quality by Design approach for analytical methods – update on recent guidelines
By Dr. Ravikrishna Chebolu
Agilent Pharmaceutical Segment Manager
Implementation of Quality by Design (QbD) principles in drug development and manufacturing has gained significant traction during the last few years. Pharmaceutical companies across the globe are in various stages of QbD implementation, and although a number of companies are still learning the art and science of QbD implementation, the industry has widely accepted its importance.
The International Conference on Harmonization (ICH) has published a number of guidelines (ICH Q8-Q11) that help you understand the principles and implementation of QbD. Starting in January 2013, the United States Food and Drug Administration (U.S. FDA) mandated inclusion of QbD elements as part of ANDAs (Abbreviated New Drug Applications) for generic drug manufacturers. With this new requirement, QbD implementation has gained further momentum.
 Enlarge
Enlarge
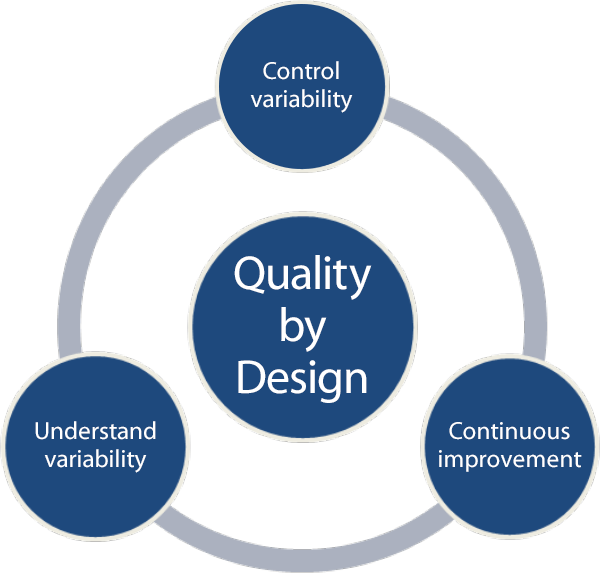
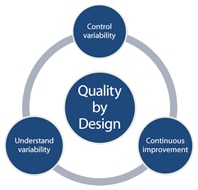
Figure 1. Key elements of QbD implementation.
Understand and control variability
The fundamental philosophy of QbD implementation requires that you understand sources of variability and that you control variability using science and risk-based methodology coupled with continuous improvement (Figure 1). You can logically apply this philosophy to any “process” (chemical, formulation, or other) in drug development and manufacturing.
A number of laboratories have started to apply principles of QbD for analytical methods and to use risk assessments and statistically designed experiments to develop robust analytical methods. Similar to the way you use QTPP (Quality Target Product Profile) and Design Space concepts for a manufacturing process, you can define ATPs (Analytical Target Profiles) and MODRs (Method Operational Design Ranges) for analytical methods.
An expert panel from the U.S. Pharmacopeial Convention (USP) published a stimuli article in July 2013 on “Lifecycle Management of Analytical Procedures.” The article suggested that you integrate traditional approaches to validation, transfer, and verification into the analytical procedure lifecycle process rather than consider them as separate entities. The expert panel listed several advantages of this approach, such as improved clarity, holistic view, and confident decision-making. In this article, ATP was identified as a fundamental component of the lifecycle approach to analytical procedures because it helps to set performance requirements for these procedures.
As part of the European Medicines Agency (EMA)-FDA pilot program for parallel assessment of QbD applications, the agencies jointly published their expectations in August 2013. They stated that “ATP can be acceptable as a qualifier of the expected method performance by analogy to QTPP as defined in ICH Q8 (R2)”, as long as the analytical methods are based on the same principles. With regard to MODR, the agencies clarified that “example data to support an MODR could include appropriately chosen experimental protocols to support proposed operating ranges/conditions and demonstration of statistical confidence throughout the MODR.”
In February 2014, FDA announced a revised draft guidance for industry titled “Analytical Procedures and Methods Validation for Drugs and Biologics” that supersedes the previous draft published in 2000. Although no specific mention of “Quality by Design” exists in this 2014 draft guidance, the terminology used throughout the document clearly points towards emphasis on QbD elements for analytical methods.
 Enlarge
Enlarge
Figure 2. Advantages of QbD approach for analytical methods.

Figure 3. Agilent primer 5991-2166EN provides examples of Agilent solutions for QbD implementation.
Important updates in latest draft guidance
When compared with the draft published in 2000, you can readily observe a couple of important updates in the 2014 draft guidance. The “Analytical methods development” section contains a recommendation to “adopt a systematic approach for method robustness study (e.g. design of experiments)” and “begin with an initial risk assessment and follow with multivariate experiments” to understand factorial parameter effects on method performance. A new section titled “Lifecycle management of analytical methods” has been added. To gain greater confidence in ensuring product quality, it discusses a continuous improvement approach for analytical methods based on new information – with the help of new technologies – related to better understanding of product critical quality attributes (CQAs).
It is relevant here to mention the ICH “Guideline for Elemental Impurities Q3D” published in 2013, which recommends a four-step process (identify, analyze, evaluate, and control) with regard to elemental impurities. The control component requires the manufacturer to test the finished product to account for any impurities that may arise during manufacturing or packaging.
In summary, a closer look at the recent guidelines published by regulatory bodies for analytical method development and validation reveals the increased emphasis on QbD principles such as science and risk based systematic approaches for robust analytical method development by understanding and eliminating the variability. More importantly, as part of lifecycle management, the guidelines encourage use of new technologies for continuous evaluation of appropriateness of analytical methods (Figure 2).
Agilent solutions aid with QbD implementation
Agilent offers tools (see Figure 3) that can help you set the design space for robust analytical methods, eliminate variability in method transfer, and evaluate CQAs for drug substances and drug products. You can combine the Agilent 1200 Infinity Series Method Development Solution with QbD software to automate the process and facilitate multivariate experiments to characterize method design space. Find more information on Agilent solutions to support QbD implementation in our recently published primer (5991-2166EN).
Note: The concepts and ideas expressed do not reflect official company policies. We recommend that all readers consult official regulatory Web sites and other reliable sources for the latest information.
>> Update My Profile | Subscribe to Access Agilent | Article Directory