Access Agilent eNewsletter, September 2013
>> Update My Profile | Subscribe to Access Agilent | Article Directory

Agilent PLgel columns provide needed sensitivity in GPC/SEC of biopharma drug excipients
By Sudharshana Seshadri and Ben MacCreath
Agilent GPC Product Managers
Excipients are cheap, inactive substances used in pharmaceutical and personal care formulations. When active ingredients (AI) are present in small quantities, excipients are used to carry the AI and make a drug tablet or capsule large enough to be easily handled. So a common painkiller could contain 80% or more inert filler. Other drug excipients ease the administration or uptake of AI, make them more palatable, or add color to aid identification. Excipients can also be used during manufacture to assist handling of the AI. For example, excipients can keep the AI from sticking to machinery or degrading during processing or storage. Excipients are also seen as influencing the rate and extent of AI uptake and are often used for this ability in time-released medications.
Many compound classes are used as excipients, including synthetic and natural polymers, saccharides, and proteins. However, there is a move away from synthetic excipients – which may be problematic for obtaining regulatory approval – and towards “natural” compounds that are potentially less toxic, more easily accessible, cheaper, and potentially more acceptable to consumers.
The value of excipients has led the biopharmaceutical industry to do extensive research in order to improve their efficacy. Because molecular weight influences many of the characteristics of excipient polymers, gel permeation chromatography/size exclusion chromatography (GPC/SEC) has become a valuable technique in this research, as you will see from the following examples.
 Enlarge
Enlarge
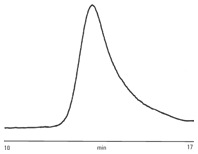
Figure 1. Analysis of polyvinylpyrrolidone using Agilent PLgel 10 µm MIXED-B columns.
Agilent PLgel 10 µm MIXED-B column successfully measures PVP in binders
Excipient binders hold tablets together. They add mechanical strength so that tablets do not disintegrate during manufacture, transport, storage, or handling. Binders also add bulk to low doses of AI.
The binder excipient polyvinylpyrrolidone (PVP) is soluble in polar organics and water. In this example, dimethyl formamide (DMF) is used with lithium bromide to minimize any polyelectrolyte effects. PVP is chromatographed successfully with Agilent PLgel 10 µm MIXED-B columns (Figure 1) on an Agilent 1260 Infinity GPC/SEC System.
Agilent PL aquagel columns reveal fine weight variations in disintegrants
Disintegrants expand and dissolve when wet, causing tablets to break apart and release the active ingredient. A disintegrant can be modified, or different disintegrants combined, so that the tablet dissolves over a period of time to deliver slow release of the active ingredient.
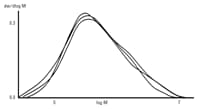
SEC reveals slight differences in the molecular size profiles of water-soluble polymers that are within the same viscosity grade. Polymers such as carboxymethyl cellulose (CMC) can have different physical characteristics due to these minute variations in the molecular weight of the material. Agilent PL aquagel-OH 40 8 µm and PL aquagel-OH 60 8 µm columns are ideal for distinguishing fine variations in CMC molecular weights because they combine low exclusion limit, high pore volume and high column efficiency (>35,000 plates/meter) for maximum resolution.
 Enlarge
Enlarge
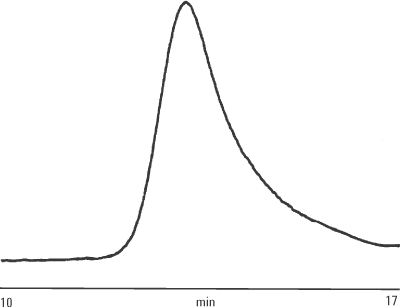
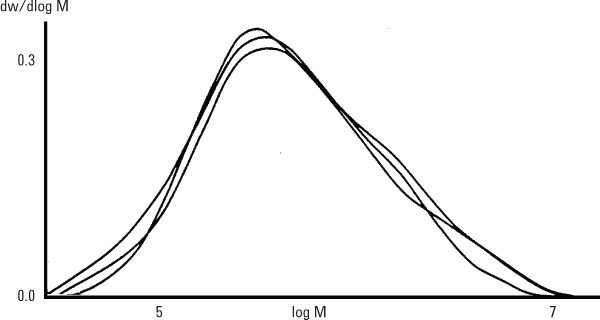
Figure 2. Raw-data chromatograms produced by Agilent PL aquagel-OH columns, show slight differences in the molecular weight of three carboxymethyl celluloses lying within the same viscosity range.
In this case, two different versions of Agilent PL aquagel-OH were connected in series to cover a molecular weight range from 104 to 107 with column calibration using Agilent pullulan standards.
Figure 2 shows the slight differences in molecular weights of three carboxymethyl celluloses that lie within the same viscosity range.
Importance of suspension and/or viscosity-increasing agents
Suspension and viscosity-increasing excipients are used to uniformly disperse other ingredients throughout a formulation, and maintain their suspension so that actives do not precipitate or settle under gravity. This is particularly valuable for liquid formulations, during and after manufacture.
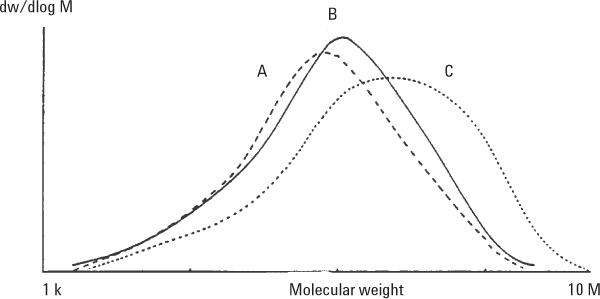
Hydroxyethyl cellulose (HEC) is widely used by the cosmetic and pharmaceutical industries as a suspending agent. You will often find it as a carrier gel for microbiocides, for example. HECs can be analyzed by aqueous GPC, but very often they are soluble in polar organic solvents, such as DMF. Agilent PLgel 5 µm MIXED-C columns are well suited to the analysis of these celluloses. Because some of these materials are ionic, a LiBr modifier is added to minimize sample aggregation (Figure 3). PEO/PEG standards are used as calibrants. Polystyrene is soluble in DMF, but some adsorption is apparent. Table 1 shows the dispersity and molecular weight averages of three HEC samples.
 Enlarge
Enlarge
Figure 3. Analysis of three HEC samples using Agilent PLgel 5 µm MIXED-C columns.
Sample |
Molecular weight average |
Polydispersity |
||
|---|---|---|---|---|
Mn |
Mw |
Mp |
||
A |
27,000 |
140,000 |
80,000 |
5.2 |
B |
30,000 |
159,000 |
102,000 |
5.2 |
C |
39,000 |
345,000 |
190,000 |
8.9 |
Table 1. Molecular weight averages and dispersity of three HEC samples
These three materials were quite different in molecular weight, indicating potential performance differences in end-use.
Trust Agilent for all your GPC/SEC needs
Agilent has a long history of involvement in the analysis of excipients by GPC/SEC, offering an array of columns, calibration standards, and instruments all developed to make your tasks easier – including Agilent PL aquagel and PLgel columns, and EasiVial and EasiCal calibration standards. Please take a few moments now to see what Agilent can do for you.
>> Update My Profile | Subscribe to Access Agilent | Article Directory
Figure 1.
Conditions |
|
|---|---|
Columns |
2 x PLgel 10 µm MIXED-B, 7.5 x 300 mm (p/n PL1110-6100) |
Eluent |
DMF + 0.1% LiBr |
Flow Rate |
1.0 mL/min |
Temp |
70 °C |
Detection |
RI |
System |
1260 Infinity GPC/SEC System |
Analysis of polyvinylpyrrolidone using Agilent PLgel 10 µm MIXED-B columns.
Figure 2.
Conditions |
|||
|---|---|---|---|
Columns |
2 x PL aquagel-OH 60 8 µm, 7.5 x 300 mm (p/n PL1149-6860) |
||
Eluent |
0.5 M Na2SO4 |
||
Flow Rate |
1.0 mL/min |
||
Detection |
RI |
System |
PL-GPC 50 Integrated System |
Raw-data chromatograms produced by Agilent PL aquagel-OH columns, show slight differences in the molecular weight of three carboxymethyl celluloses lying within the same viscosity range.