Access Agilent eNewsletter, July 2013
>> Update My Profile | Subscribe to Access Agilent | Article Directory

Particle trapping technology delivers the benefits of PLOT columns to GC/MS
By Yun Zou
Agilent GC Applications Chemist
Because particle shedding of the stationary phase can make it challenging to use porous layer open tubular (PLOT) columns, traditional PLOT columns are seldom used for GC/MS. When particles dislodge from the column and flow downstream, they can either restrict the flow or plug the GC flow path causing damage to the column switching valves, as well as detector contamination. If particles enter the detector, the resulting signal spikes can interfere with the software’s ability to identify and quantitate compounds.
Some labs have attempted to solve the problem of particle shedding with inconvenient workarounds, such as installing a particle trap – via press fit or other union and associated fittings – that can clog. Others use inline frits on the column at the valve or detector, which require periodic maintenance.
PLOT PT columns end the problems caused by particle shedding
Now, Agilent J&W PLOT PT columns put an end to problems caused by particle shedding thanks to an integrated particle trapping technology on both ends of each column – which enables worry-free GC/MS detection and enhanced analytical capability for hydrocarbon processing industry (HPI) applications.
In this article, we discuss two HPI applications that illustrate the advantages of using Agilent J&W PLOT PT columns and GC/MS when the matrix is quite complex and can interfere with the quantitation of the target compounds by GC/FID or GC/TCD. The first example evaluates using GC/MS with an Agilent J&W PLOT PT column for coal-to-chemical process gas analysis and the second evaluates using an Agilent J&W PoraBOND Q PT column for oxygenates in mixed C4 hydrocarbon streams analysis.
 Enlarge
Enlarge
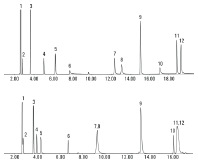
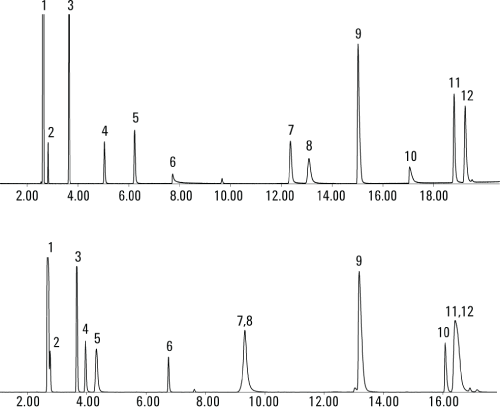
Figure 1. TIC of gas mix using an Agilent GC/MSD system and an Agilent J&W HP-PLOT Q PT column (above) and HP-PLOT U PT column (below).
Improving peak shape and resolution for coal-to-chemical process gas analysis
As oil prices continue to rise, the chemical industry is finding coal to be an attractive feedstock, especially in countries where it is abundant. The high demand for propylene and ethylene are driving interest in coal-to-olefins (CTO) processes. In a typical CTO process, methanol from coal or natural gas is first dehydrated to dimethyl ether (DME). Then the equilibrium mixture is converted to light olefins through a complex network of chemical reactions. Right now researchers are focused on developing high-efficiency catalysts and optimizing process conditions that can improve olefin yield, and analyzing the components in coal-to-chemical process gas by separating polar and nonpolar compounds – which is very important for this research.
Figure 1 shows Agilent J&W HP-PLOT Q PT and HP-PLOT U PT columns used for typical coal-to-chemical process gas analysis with GC/MS. The HP-PLOT U PT column is a more polar phase, so it demonstrates improved inertness and better peak shape and response for very polar compounds such as methanol and hydrogen sulfide. This enables lower detection limits for these compounds. However, there are two pairs of coeluting compounds on the column, namely propylene and propane, and 2-butylene and butane. In this particular instance, the HP-PLOT Q PT column improves the separation of polar and nonpolar compounds and provides better resolution of hydrocarbon isomers as detailed in Application Note 5991-2205EN.
Accurately monitor impurities in oxygenates in mixed C4 hydrocarbon streams
C4 hydrocarbons such as butadiene, butenes and butanes are used as feedstock for industrial chemicals, rubber, and plastics. They are produced by a steam cracking process. However, due to the presence of harmful impurities in mixed C4 streams, only a small proportion of valuable components are extracted and subsequently processed into usable products. Most C4 by-products are flared or can only be used as low-quality, low-value additives.
Most petrochemical companies are very interested in maximizing the mixed C4 stream’s yield of high-quality components and turning them into value-added products. The key to this objective is accurately monitoring the impurities.
One effective way to improve the value of mixed C4 is to produce propylene using the mechanism of 2-butene and ethylene disproportionation. This technology can be used with a variety of C4 streams, including the mixed C4 produced by steam cracking, raffinate C4 mixtures from methyl tertiary butyl ether (MTBE), or butadiene extraction. Fresh C4, plus C4 recycle, are mixed with ethylene and sent through a guard bed to remove trace impurities – including DME, methanol (MeOH), ethanol (EtOH), MTBE, and ethyl tertiary butyl ether (ETBE) – from the mixed feed. It is crucial that these impurities are thoroughly monitored because trace oxygenates can lead to catalyst poisoning, which halts reactions or inhibits yields.
 Enlarge
Enlarge
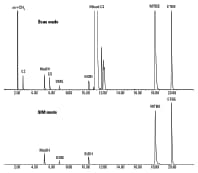
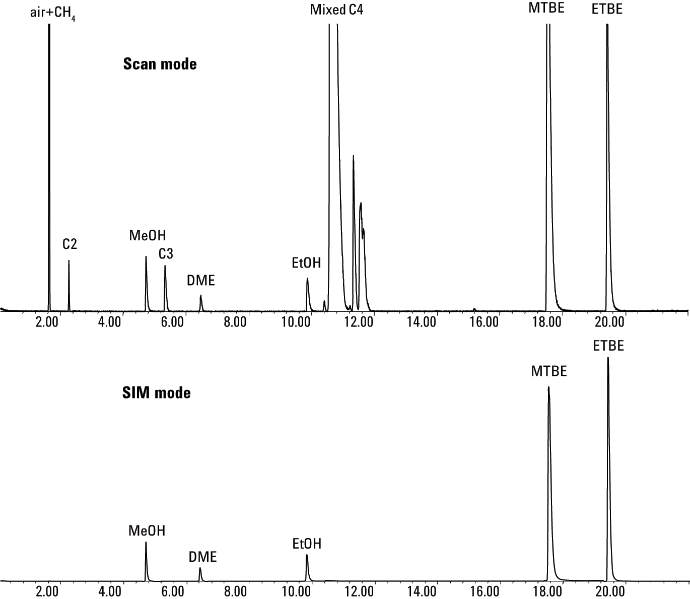
Figure 2. Simultaneous scan (top) and SIM (bottom) analysis of raffinate C4 mixtures from MTBE using an Agilent GC/MS system and an Agilent J&W PoraBOND Q PT column.
One approach is to use GC/FID and choose a selective column that promotes interference-free elution of oxygenates from hydrocarbons. A better solution is to use of highly sensitive and selective GC/MS for identification purposes, because this avoids the detection interference of hydrocarbons.
Figure 2 shows the total ion chromatogram of raffinate C4 mixtures from MTBE. Synchronous SIM/scan is used to monitor ions of interest with high-sensitivity SIM mode and to simultaneously acquire library-searchable scan data in one run. In SIM mode, the method can eliminate air and hydrocarbon interferences to allow for MS identification of the target oxygenates and achieve reliable analysis as demonstrated in Application Note 5991-1549EN.
PLOT a route to more accurate analysis and greater efficiency
If you use PLOT columns with MS detection and capillary flow technology, and want to avoid problems with unions or downstream filters, Agilent PLOT PT columns with integrated particle trapping technology provide the enhanced analytical capability you need. Take a moment now to learn more about how Agilent PLOT PT columns provide more confident identifications of unknowns and take the worry out of performing GC or GC/MS analysis on a day-to-day basis. Then explore our next generation Agilent 5977A MSD – the latest addition to the most popular GC/MS of all time.
>> Update My Profile | Subscribe to Access Agilent | Article Directory
Figure 1.
Conditions |
|
|---|---|
Columns |
Agilent J&W HP-PLOT Q PT, 30 m x 0.32 mm, 20 µm (p/n 19091P-QO4PT) |
Carrier |
Helium, constant flow mode, 35 cm/s, 32 °C |
Oven |
32 °C for 5 min, 32 °C to 70 °C at 30 °C/min, 70 °C for 5 min, 70 to 160 °C at 10 °C/min |
Injection |
170 °C, split ratio 5:1, 250 µL gas sampling loop |
Instrument |
Agilent 7890A GC with gas blending system |
MS |
EI, Scan/SIM |
Transfer line |
180 °C |
MS temperature |
230 °C (source), 150 °C (quad) |
Scan mode |
Mass range (10 to 100 amu) |
Peak ID
- CO/air
- Methane
- Carbon dioxide
- Ethylene
- Ethane
- Hydrogen sulfide
- Propylene
- Propane
- Dimethyl ether
- Methanol
- Butylene
- Butane
TIC of gas mix using an Agilent GC/MSD system and an Agilent J&W HP-PLOT Q PT column (above) and HP-PLOT U PT column (below).
Figure 2.
Conditions |
|||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Column |
Agilent J&W PoraBOND Q PT, 30 m x 0.32 mm, 5 µm (p/n CP7351PT) |
||||||||||||||||||||||||||||||
Carrier |
Helium, constant flow mode, 39 cm/s, 48 °C |
||||||||||||||||||||||||||||||
Oven |
48 to 90 °C at 6 °C/min, 90 to 240 °C at 15 °C/min, 240 °C for 10 min |
||||||||||||||||||||||||||||||
Injection |
200 °C, split ratio 5:1, 200 µL gas sampling valve |
||||||||||||||||||||||||||||||
GC |
Agilent 7890A Series GC |
||||||||||||||||||||||||||||||
MS |
EI, Scan/SIM |
||||||||||||||||||||||||||||||
Transfer line |
280 °C |
||||||||||||||||||||||||||||||
MS temp |
230 °C (source), 150 °C (quad) |
||||||||||||||||||||||||||||||
Scan mode |
Mass range (10 to 200 amu) |
||||||||||||||||||||||||||||||
SIM mode |
|
||||||||||||||||||||||||||||||
Simultaneous scan (top) and SIM (bottom) analysis of raffinate C4 mixtures from MTBE using an Agilent GC/MS system and an Agilent J&W PoraBOND Q PT column.