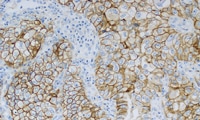
In NSCLC, PD-L1 testing with PD-L1 IHC 22C3 pharmDx can help identify patients for first-line treatment with KEYTRUDA
* Review the PD-L1 IHC 22C3 pharmDx Instructions for Use for more information regarding the KEYNOTE clinical trials
- Lung cancer is the leading cause of cancer-related mortality in the United States, and NSCLC accounts for 85% of all lung cancer cases
- In 2016, KEYTRUDA became the first anti-PD-1 monotherapy FDA approved at first line for patients with metastatic NSCLC
- PD-L1 testing provides a direct assessment of PD-L1 expression, which is a biomarker for response to anti-PD-1 therapy in NSCLC
- PD-L1 testing with PD-L1 IHC 22C3 pharmDx was used to qualify patients with NSCLC for first-line treatment with KEYTRUDA in the KEYNOTE-024 and KEYNOTE-042 clinical trials*
The only PD-L1 assay used in the KEYTRUDA KEYNOTE-042 clinical trial in NSCLC
PD-L1 IHC 22C3 pharmDx is the FDA-approved companion diagnostic for KEYTRUDA, and was used to assess PD-L1 expression and select patients for treatment in KEYNOTE-042

† Percentage calculation based on patients whose tumors expressed PD-L1 (TPS ≥ 1%). Patients whose tumors did not express PD-L1 were not enrolled
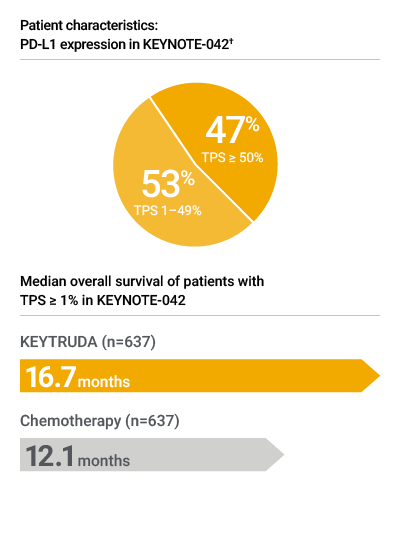
When confidence in a PD-L1 test is critical, the ONE you choose is crucial
- The ONE PD-L1 assay used in KEYTRUDA clinical trials1,2
- The ONE PD-L1 assay first approved with KEYTRUDA in every indication that requires PD-L1 testing1,2
- The ONE PD-L1 assay trusted worldwide to test hundreds of thousands of patients for KEYTRUDA9

KEYTRUDA® is a registered trademark of Merck Sharp & Dohme LLC, a subsidiary of Merck & Co., Inc., Rahway, NJ, USA
References: 1. PD-L1 IHC 22C3 pharmDx [package insert]. Carpinteria, CA: Dako, Agilent Pathology Solutions; 2019. 2. Keytruda [package insert]. Kenilworth, NJ: Merck & Co., Inc.; 2019. 3. Noone AM, Howlader N, Krapcho M, et al. SEER Cancer Statistics Review, 1975-2015. National Cancer Institute. https://seer.cancer.gov/csr/1975_2015/. November 2017 SEER data submission. Published April 2018. Accessed September 21, 2018. 4. Molina JR, Yang P, Cassivi SD, Schild SE, Adjei AA. Non-small cell lung cancer: Epidemiology, risk factors, treatment, and survivorship. Mayo Clin Proc. 2008;83(5):584-594. 5. FDA Approves Merck’s KEYTRUDA® (pembrolizumab) in Metastatic NSCLC for First-Line Treatment of Patients Whose Tumors Have High PD-L1 Expression (Tumor Proportion Score [TPS] of 50 Percent or More) With No EGFR or ALK Genomic Tumor Aberrations; October 24, 2016. https://investors.merck.com/news/press-release-details/2016/FDA-Approves-Mercks-KEYTRUDA-pembrolizumab-in-Metastatic-NSCLC-for-First-Line-Treatment-of-Patients-Whose-Tumors-Have-High-PD-L1-Expression-Tumor-Proportion-Score-TPS-of-50-Percent-or-More-With-No-EGFR-or-ALK-Genomic-Tumor-Aberrations/default.aspx. Accessed March 22, 2019. 6. Brahmer JR, Govindan R, Anders RA, et al. The Society for Immunotherapy of Cancer consensus statement on immunotherapy for the treatment of non-small cell lung cancer (NSCLC). J Immunother Cancer. 2018;6(1):75. 7. Hanna N, Johnson D, Temin S, et al. Systemic therapy for stage IV non–small-cell lung cancer: American Society of Clinical Oncology Clinical Practice Guideline Update. J Clin Oncol. 2017;35(30):3484-3515. 8. Planchard D, Popat S, Kerr K, et al. Metastatic non-small cell lung cancer: ESMO Clinical Practice Guidelines for diagnosis, treatment and follow-up. Annals of Onc. 2018;29(4):iv192-iv237. 9. Data on file. Agilent Technologies, Inc.
For countries outside of the United States, see the local KEYTRUDA product label for approved indications and expression cutoff values to guide therapy.
D51249/02.1