PD-L1 IHC 28-8 pharmDx for Non Small Cell Lung Cancer


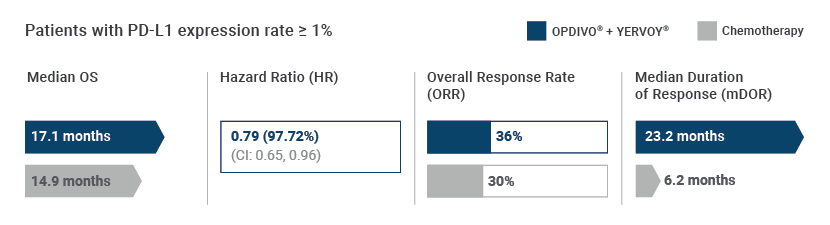
CHECKMATE-227 was a Phase III, randomized, multi-center, multi-cohort, open-label study in patients who had no prior anti cancer therapy with metastatic NSCLC. Part 1a of the study investigated patients with PD-L1 expression level ≥ 1%, who were previously untreated for advanced disease. PD-L1 IHC 28-8 pharmDx was the only test used for PD-L1 expression analysis across all histology in non-squamous and squamous NSCLC.
CHECKMATE-227, Part 1a highlighted the significant survival and response outcomes from combining OPDIVO® (nivolumab) plus YERVOY® (ipilimumab) as a first line treatment for metastatic NSCLC patients whose tumors exhibited PD-L1 expression ≥ 1%.
* PD-L1 IHC pharmDx is for in vitro diagnostic use.
PD-L1 IHC 28-8 pharmDx is FDA-approved and fully validated with analytical performance having met all pre-determined acceptance criteria for sensitivity, specificity and precision.
| Selected analytical validation parameters | Description |
|---|---|
| Specificity |
|
| Sensitivity |
|
| Repeatability |
|
| Reproducibility |
|
OA = Overall Agreement
| Product | Code |
|---|---|
| PD-L1 IHC 28-8 pharmDx | SK005 |
|
Required but not included in kit: Autostainer Link 48 EnVision FLEX Wash Buffer, 20x EnVision FLEX Hematoxylin (Link) PT Link PT Link Rinse Station |
AS480 K8007 K8008 PT101 / PT200 PT109 |
- CHECKMATE-227. M.D. Hellmann, et al. Nivolumab plus Ipilimumab in Advanced Non–Small-Cell Lung Cancer, The New England Journal of Medicine, 2019.
- PD-L1 IHC 28-8 pharmDx Instructions for Use.
- YERVOY® package insert. Manufactured by: Bristol-Myers Squibb Company, Princeton, NJ 08543 USA U.S. License No. 1713.
- OPDIVO® package insert. Manufactured by: Bristol-Myers Squibb Company, Princeton, NJ 08543 USA U.S. License No. 1713.
Intended Use
For in vitro diagnostic use.
PD-L1 IHC 28-8 pharmDx is a qualitative immunohistochemical assay using Monoclonal Rabbit Anti-PD-L1, Clone 28-8 intended for use in the detection of PD-L1 protein in formalin-fixed, paraffin-embedded (FFPE) non-small cell lung cancer (NSCLC), squamous cell carcinoma of the head and neck (SCCHN), and urothelial carcinoma (UC) tissues using EnVision FLEX visualization system on Autostainer Link 48.
PD-L1 protein expression is defined as the percentage of evaluable tumor cells exhibiting partial or complete membrane staining at any intensity.
Companion Diagnostic Indication
|
Tumor Indication |
PD-L1 Expression Clinical |
Intended Use |
|
NSCLC |
≥ 1% tumor cell expression |
PD-L1 IHC 28-8 pharmDx is indicated as an aid in identifying NSCLC patients for treatment with OPDIVO® (nivolumab) in combination with YERVOY® (ipilimumab). |
PD-L1 expression (≥ 1% or ≥ 5% or ≥ 10% tumor cell expression), as detected by PD-L1 IHC 28-8 pharmDx in non-squamous NSCLC may be associated with enhanced survival from OPDIVO®.
PD-L1 expression (≥ 1% tumor cell expression), as detected by PD-L1 IHC 28-8 pharmDx in SCCHN may be associated with enhanced survival from OPDIVO®.
PD-L1 expression (≥ 1% tumor cell expression), as detected by PD-L1 IHC 28-8 pharmDx in UC may be associated with enhanced response rate from OPDIVO®.
See the OPDIVO® and YERVOY® product labels for specific clinical circumstances guiding PD-L1 testing.




