TRS100 定量药物分析系统
产品详细信息
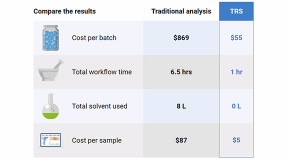
- 检测数百个片剂或胶囊,几分钟内即可完成。
- 适用于完整胶囊和大多数厚度范围的包衣片的分析。
- 无需样品前处理、湿化学法或熟练的测试人员。
- 一次测量即可实现 APIs 和赋形剂的定量分析。
- 定量限低至 0.1% w/w。
- 定量分析完整片剂中的多晶型。
- 精益校准模型,易于创建和维护。
- 方法已得到监管部门批准。
- 提供适用于片剂、胶囊、液体和粉末的各种样品盘和附件。
- 内置的技术控制功能可确保数据安全并控制访问,帮助您遵循 US FDA 21 CFR Part 11 和欧盟附录 11 的要求。
- 可持续性之选:TRS100 经过独立审计,确认其在整个产品生命周期对环境的影响后,获得了 My Green Lab 的 ACT(归责性、一致性、透明度)标签。
| 光谱范围 |
|
| 校准标样 |
|
| 样品瓶类型(可选) |
|
| 法规认证 |
|
| 激光波长 |
|
- 重要资料
-
Implementing Transmission Raman Spectroscopy for Fast Content Uniformity Testing: From Feasibility Evaluation to a Validated Release Method
Dr. Meike Romer discusses why transmission Raman spectroscopy is ideal for content uniformity, how it works in conjunction with established liquid chromatography (LC) methods, and the approach that led to the implementation of TRS systems for content uniformity testing at Grünenthal Pharma.
- 白皮书
- English
- 2021 年 3 月 30 日
- 1.46 MB
Quantitative Determination of Salt-Form Impurities of Warfarin at the Lowest Commercial Dose Strength using Transmission Raman Spectroscopy
An analysis and quantification of Warfarin sodium salts in intact tablets at low doses.
- 应用简报
- English
- 2019 年 5 月 20 日
- 1.66 MB
Quantification of Tablets Containing Multiple APIs Using Transmission Raman
The quantification of three active pharmaceutical ingredients and 2 excipients using a 9 second transmission Raman spectroscopy measurement
- 应用简报
- English
- 2018 年 1 月 22 日
- 788.16 KB
- 应用简报
- 产品样本
- 宣传单页
- 基础导论
- 白皮书
- 常见问题解答
-
TRS100 和透射拉曼光谱常见问题解答
使用透射拉曼光谱 (TRS) 和 Agilent TRS100 进行含量均一度检测、多晶型分析和结晶度分析的常见问题解答。了解 TRS 技术基础知识、方法开发、制药 QC 成本节省、产品适用性、监管环境等更多信息。
- 现场准备清单
- 用户手册
视频
Read reviews on the TRS100 Raman pharmaceutical analysis system
Explore customer reviews posted on SelectScience, or write your own review.
Raman Spectroscopy Applications Guide
A guide to Raman spectroscopy applications using Agilent handheld and benchtop Raman analyzers.
安捷伦透射拉曼光谱助力口服固体药物的连续生产
本应用简报介绍了透射拉曼光谱 (TRS) 作为一种近线定量分析技术在口服固体制剂连续生产中的应用。TRS 可对片剂和胶囊进行快速、高通量、近乎实时的整体分析。
- 2025 年 7 月 2 日
已开放注册。2024 年传输拉曼光谱虚拟用户组会议
与来自世界各地的透射拉曼光谱 (TRS) 爱好者一起加入我们。在本次活动中,我们将探索 TRS 技术激动人心的领域及其成功应用于制药应用中的批量定量分析。了解 TRS 如何克服传统拉曼光谱的局限性,实现对完整药物片剂和胶囊的快速非侵入性批量分析。立即注册!
- 2024 年 9 月 29 日
Agilent TRS100 获得 My Green Lab 的 ACT 认证标签
Agilent TRS100 拉曼光谱系统已通过欧盟、英国和美国的 ACT 标签认证。这一关于系统环境影响的明确第三方验证信息使实验室能够轻松选择有助于其实现可持续发展目标的产品。
- 2024 年 3 月 31 日
Jerry Jin
PAT Scientist at Allergan
The analysis of 10 pharmaceutical tablets for their content uniformity takes only several minutes, as compared to one day turnaround time when using a traditional HPLC method. Method development using TRS100 for material identification and quantitative assay is generally easier than other vibrational spectroscopic techniques such as near infrared (NIR). One intrinsic advantage of TRS is its excellent specificity. Considering the unique strength of Agilent’s TRS100 in pharmaceutical analysis, I expect to see its deployment in more QC labs for product identification and assay, as well as its deployment on factory floors for real-time release testing.
查看更多使用透射拉曼光谱快速进行含量均一度检测 — 从可行性评估到经验证的放行方法
本次网络研讨会着重介绍了使制药公司决定采用透射拉曼光谱系统进行含量均一度检测的方法。此外还将讨论选择透射拉曼光谱进行含量均一度检测的原因以及其如何与现有的液相色谱 (LC) 方法配套使用。
使用透射拉曼光谱扩展药物片剂剂量单位测试的均一度
药物制剂开发中的固态分析:透射拉曼光谱和 LDIR 激光红外成像技术的优势和局限
透射拉曼光谱 (TRS) 已获得制药行业应用批准,可用于散装原辅料样品的分析检测和鉴定以及成品含量均一度测试。本网络研讨会的第一部分将介绍拉曼光谱的基本原理、拉曼光谱与传统测试方法相比的优势和局限性、选择适合拉曼检测的产品所需的流程和步骤,以及拉曼方法和系统的开发和验证要求。此外,还将概述拉曼光谱方法的申报要求以及已获批的拉曼系统的生命周期维护。本网络研讨会的第二部分将介绍 LDIR 技术如何对较大的片剂表面进行快速、高质量的表面成像,应用范围包括制剂开发中的多晶型筛选。
- 出版物
-