The "green rush" is far from over. Legalization of cannabis continues to expand throughout the United States. According to recent research, the cannabidiol (CBD) market is poised to grow from just over $2 billion USD in 2019 to $39 billion by 2027, with a compound annual growth rate (CAGR) of 46.4% during that period.1 The market trajectory remains positive in the face of downward price pressure due to commoditization, and despite the coronavirus pandemic (cannabis enterprises are widely designated as essential businesses).
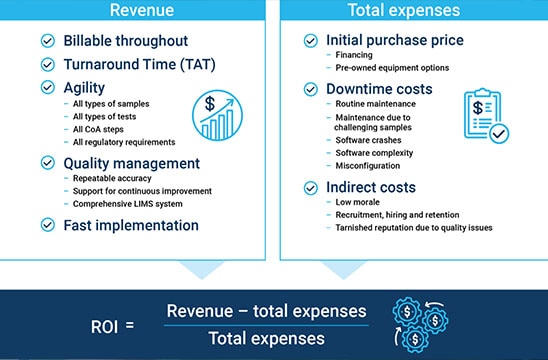
So, what is the best way for your lab to maximize return on investment—with minimal cost, complexity, and risk? Clearly, this is no small undertaking. Setting up a profitable cannabis testing laboratory requires a suite of analytical tools, expertise in multiple facets of chemical and biological testing, continuous compliance with everchanging regulations, and the business acumen to plan and implement the right strategy. The one element that seems relatively simple, at least on the surface, is calculating the expected return on investment (ROI) for cannabis testing. The ROI equation is straightforward:

The question is, are you accurately accounting for everything that influences revenue and expenses? The ROI equation expands into multiple factors—some more obvious than others. Capital investment—the price tag of your cannabis testing equipment—is one of the largest expenses, but your choice of a vendor will impact other major expenses. All too often, newcomers to the market overlook direct and indirect costs that impact ROI. This can lead to the wrong "go/no-go" decision, delayed time to market, extremely disappointing ROI results, or unanticipated losses even in the face of a thriving, high-growth market. This article examines hidden costs and factors to consider when formulating a business plan from a meaningful perspective: reality. Here are seven key attributes of analytical instruments, software, and services you should consider as you evaluate cannabis testing equipment options with the goal of maximizing your ROI.
#1: Billable throughput
The speed at which your lab instruments can process samples is the primary gating factor for revenue, and that is why total testing capacity is a commonly used metric for estimating revenue. However, that approach can lead to overly optimistic ROI results. A more accurate and meaningful metric, in terms of revenue, is Turnaround Time (TAT).
The reason: TAT highlights the rate-limiting step in your process—and underscores the crucial difference in ROI between total capacity and billable throughput. For example, in many cases producers require seven cannabis assays (potency, terpenes, heavy metals, microbial, residual solvents, pesticides, and mycotoxins), and they can only be billed once all seven are completed. If your lab can analyze 400 samples for heavy metals on an ICP-MS2 in a day but can only test about 20 samples for pesticides per day with an LC-MS/MS system, the billable throughput is bottlenecked to 20 customer samples per day, not 400. Moreover, a single grower's lot could take up the whole 20-sample sequence, which limits throughput and the ability to serve a diverse customer base—impacting revenue both directly and indirectly. Given the scenario above, it would be wise to discuss higher-throughput instrument options with your vendors.
#2: Multi-dimensional agility
Once you understand that TAT, not total capacity, is the more accurate way to forecast revenue, it becomes clear that many other factors also influence TAT. These factors could all be categorized under the umbrella of "agility," which should include:
- The ability to handle multiple types of samples (oils, tinctures, concentrates, edibles, and beverages) from multiple sources (growers, extractors, manufacturers, etc.).
- The flexibility to perform multiple types of tests (pesticides and mycotoxins, potency, terpenes, heavy metals, microbial testing, residual solvents) and adapt those tests as customer or regulatory requirements change. For example, pesticide analysis requirements are likely to change with evolving regulations.
- The ability to process all steps in the Certificate of Analysis (CoA) process: Sample reception, sample preparation, data acquisition, data analysis, generating the data package required for the CoA.
- The ability to meet compliance requirements for a wide range of fast-changing regulations. Delivering all the attributes of agility gives your laboratory a distinct competitive advantage, while gaps in any of these areas can lead to paying more and earning less.
In this context, the advantages of a multi-instrument approach become starkly clear. A lab that harnesses both LC-MS/MS and GC-MS/MS instruments for pesticides and mycotoxin analyses in regions where this is required can dramatically increase TAT, which means higher billable throughput. For example, one vendor's single platform approach of LC-MS/MS can only process about "24 vials" per day,3 whereas a dual-instrument approach can support 80 – 85 billable samples per day.4,5 The multi-instrument approach also contributes to sustainably high ROI through ongoing competitive advantages and a more diverse base of potential customers.
#3: Comprehensive quality management
Virtually all analytical labs take quality very seriously and invest heavily in quality management. However, many do not fully consider the link between quality management and ROI—perhaps because quality is a business imperative while ROI is a business consideration. However, there need not be a trade-off. In fact, the highest quality programs are likely to improve ROI. Therefore, rather than view quality as a cost center for the lab or a regulatory requirement, it pays to view quality management and assurance as a major opportunity for raising ROI and the reputation of your lab. The selection of a vendor and lab equipment for cannabis testing can contribute to more comprehensive quality management and ROI in several ways.
For example, the right equipment can foster repeatable accuracy and precision in results, leading to a reputation for quality which is likely to result in a larger customer base. In addition, a vendor's ability to improve every aspect of quality management that impacts ROI—from a focus on TAT, to method development, to excellent staff training, to reagent preparation—is critical. Simply put, by not compromising on these principles, laboratories can minimize the risk of errors, strengthen their position in the industry, elevate their reputation among potential customers, and maximize ROI.
#4: Fast implementation timeframe
Revenue projections start with the assumption that the equipment is operating properly and meeting all specifications. The question you need to ask prospective vendors: how long will that take? There are several factors that can delay or complicate the implementation timeframe, including:
- The availability of excellent service and support in installing equipment. Don't take this for granted. Vendors vary widely in their ability to send the right experts with the right implementation plan to your site in a timely and cost-effective way. In addition, instruments vary widely in complexity, number of components, steps in implementation, and other factors that impact your implementation timeframe.
- Effective, affordable training and education for staff, including learning how to use the software. If the user interface is non-intuitive or difficult to learn; if training materials are poorly made; if instructors have insufficient knowledge or experience; or if it takes excessive time to get answers to technical questions once the instructors leave, ROI (and morale) will suffer.
- Development and implementation of analytical methods that consistently comply with stringent regulations. To maximize ROI, this would include ready-to-run, easily downloadable digital information packages that provide everything needed to set up a complete testing workflow. The methods packages should be flexible enough to meet your specific testing needs, support a tandem instrument approach, and supply you with:
- Guidance on the most suitable instrument configuration for your workflow
- Optimized analytical methods for sample introduction, chromatographic separation, detection, and data analysis
- Sample preparation protocols and consumables
- Best practices for implementation across workflows, including potency, pesticide residues and mycotoxins, heavy metals, residual solvents, and terpenes.
5: Affordability
The term "affordability" has multiple shades of meaning, triggering several important questions to consider.
For example:
- Does the purchase price of the cannabis testing equipment allow for high, sustainable ROI? A small price tag does not necessarily translate to higher ROI. Again, the key metric to consider is TAT. If the equipment supports a combination of high billable throughput, a fast implementation timeframe, reliably high-quality sample testing, and minimal downtime and maintenance requirements, a high purchase price may lead to much higher ROI than the low-cost alternative.
- What options are available for attaining the capital required for purchase? Drawing down the lab's capital assets or seeking more venture capital are not the only options for securing funding for new lab equipment. Look for vendor-supported financing and/or pre-owned equipment options offered by the vendor to help cut the buying cycle, get up-and-running quickly, and reduce the initial financial outlay.
- What is the total cost of ownership? A low purchase price may mask significant costs such as consulting fees to implement and maintain the equipment, instrument downtime (see below), education and training requirements, lack of flexibility/agility in methods testing capabilities, and more.
Single-platform or dual-platform approach?
Do the math.
When you consider the actual costs and expected revenue of single-platform vs. dual-platform solutions, the ROI results may surprise you. Consider a realistic example: Typically, a full analytical instrument suite for a cannabis testing lab costs in the range of $800,000 USD for LC-MS/MS only, and approximately $1,200,000 for a dual instrument approach in regions where this is required.
However, if your lab will analyze more than 16 samples per day, the costs for the LC-MS/MS-only approach could exceed $2,000,000 due to the need for more systems. With limited funds and the need to conserve operational capital, the lower-cost option is tempting. However, this decision can lead to lower ROI.
With LC-MS/MS only, a cannabis lab at full operational efficiency can typically produce a maximum theoretical output of 24 injections per 24-hour period due to the analysis method consuming more than 40 minutes (see Footnote 3). Vendors that advocate for LC-MS/MS-only have revealed that it typically takes a minimum of 40 minutes to analyze pesticides and mycotoxins because each sample must be run twice via two distinctly different methods. As noted above, this results in about 14 – 16 billable samples per day. By using both a GC-MS/MS and an LC-MS/MS in regions where this is required due to inclusion of certain pesticides in the target list, the lab has more than 5 – 6 times the sample capacity compared to a single-platform approach. If the cannabis testing lab is charging $500/sample, the LC-MS/MS-only approach, at 16 samples/day caps the revenue at $8,000/day. A dual instrument approach running 80 samples a day can generate $40,000/day.
Excluding labor costs and overhead, recovering the initial investment for lower-priced approaches (an $800,000 price tag) could take approximately 64 fully operational days, whereas with the dual-instrument approach this could be realized in just 30 days.
#6: Downtime costs
Equipment t downtime adds to the lab's operating costs because it interrupts sample analysis, impacts productivity, and requires repairs or upgrades. Your ROI projections should consider the many causes and timeframes of downtime, including:
- Routine maintenance: Every piece of equipment t requires occasional maintenance, but some require that maintenance be performed only by highly skilled and expensive third-party support specialists. Make sure you know and understand the financial impact of all "routine" maintenance.
- Added maintenance requirements due to challenging samples. Instrument maintenance may be needed due to contamination from poor sample preparation. Samples that are "winterized" or simply diluted before analysis (with solvents like acetonitrile) quickly foul and dirty the injector and source regions of the analytical instrument. This downtime translates to lost revenue and can negate any initial savings of cheaper sample preparation methodologies.
- Software crashes: Instruments depend on software, and software varies widely in its uptime. Crashes, frequent update requirements, or extremely complex maintenance can create unexpected expense and lost productivity.
- Extreme software complexity and non-intuitive interfaces can lead to high training and education costs, as well as downtime due to user errors.
- Poorly configured analytical systems can lead to reduced billable throughput, higher TAT, and additional costs for consultants and specialists to correct the configuration.
#7: Indirect costs
"Ultimately, instrument selection should not be about cost alone; you must consider cost of ownership and the support offered by the vendor. Servicing and the availability of practical support are key to minimizing instrument downtime. Every day lost while waiting for the support you need to reach your lab is bad for business and bad for your customers."
– Swetha Kaul, Ph.D., Board Vice President, California Cannabis Industry
Immediate capital expenditures are only one element of total expense. It is important to also consider the non-tangible, long-term costs of your instruments.
- Low morale due to overburdened staff. This can be the direct result of excessively complex or difficult to use equipment, but it has indirect costs in the form of higher labor expense and more downtime. In addition, low morale leads to faster employee turnover and reduced ability to attract and retain top talent, which in turn creates higher recruitment costs and salary requirements.
- Any of the factors that contribute to low morale can also result in damage to the lab's reputation, which in turn leads to a reduction in TAT, lower total revenue, and higher total expense.
- Lower-quality test results are ultimately the byproduct of the wrong equipment choice, and the indirect costs include permanent damage to the lab's reputation and an inability to keep excellent employees on staff.
About Agilent
Agilent has developed a total, trusted package of analytical instruments and methodologies that address every aspect that impacts the ROI of cannabis testing.
We offer:
- Robust instruments designed for productivity, software, services, consumables, and sample preparation. For details visit https://www.agilent.com/en/solutions/cannabis-hemp-testing
- A dual-instrument approach for pesticide and mycotoxin analyses that maximizes agility and TAT, proven at multiple cannabis testing laboratories worldwide. For an example, read about the experiences of Cambium Analytica at: Cambium Analytica
- Simplify and faster setup and training eMethods and a team of cannabis application experts ready to help develop, implement, and optimize methods to accelerate your laboratory success. For details visit https://explore.agilent.com/cannabis-testingemethods
- Save time and simplify decision making with Cannabis consumable kits and analytical products. Learn more at: https://explore.agilent.com/Cannabis-kits#
- A single trusted source for your entire cannabis testing workflow. For additional information, email us at cannabis.inquiries@agilent.com or contact us directly at the numbers below.
Contributors:
Greg Kozadjian, Agilent Technologies
Anthony Macherone, Ph.D, Agilent Technologies
Contact us:
U.S. and Canada
1-800-227-9770
agilent_inquiries@agilent.comEurope
agilent_inquiries@agilent.comAsia Pacific
inquiry_lsca@agilent.com
- Fortune Business Insights report, "Cannabidiol (CBD) Market Size, Share & COVID-19 Impact Analysis, 2020-2027."
- Craig Jones, Jenny Nelson. (2017) Multi-Element Analysis of Cannabis using the Agilent 7800 ICP-MS. Agilent Application Note 5991-8482. © Agilent Technologies, Inc.
- Rhoden, T. (2020) Setting Up a Successful Cannabis Lab - It's All in the Details. Retrieved October 27, 2020 from https://global.gotowebinar.com/join/3339242908 01355789/779531168.
- Jeffery S. Hollis, Eric Fausett, Jessica Westland, Anthony Macherone. (2019) Analysis of Challenging Pesticides Regulated in the Cannabis and Hemp Industry with the Agilent Intuvo 9000‑7010 GC/MS/MS system: The Fast-5. Agilent Application Note 5994-1604. © Agilent Technologies, Inc.;
- Peter J.W. Stone, Jennifer Hitchcock, Jean‑Francois Roy, Christophe Deckers. (2020) Determination of Pesticides and Mycotoxins in Cannabis Flower as Defined by Legalized U.S. State Recreational Cannabis Regulations. Agilent Application
Related Content:
First AOAC Approved Method for Heavy Metals Testing in Cannabis and Cannabis products
Cannabis potency testing methodologies: What are the options?
How to Select the Right Equipment for Cannabis Potency Testing
Common Pitfalls in Cannabis Lab Testing: What You Should Know and How to Avoid Them