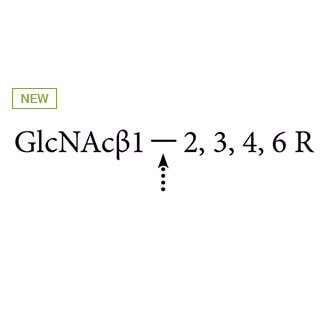
Specifications
| Concentration |
|
| Enzyme Applications |
|
| Enzyme Formulation |
|
| Enzyme Source |
|
| Enzyme Specificity |
|
| Enzyme Unit Definition |
|
| Volume |
|
| pH Optimum |
|
Support
- Safety Data Sheets
-
AdvanceBio β-N-Acetylhexosaminidase, 5x Reaction Buffer A
Material Safety Data Sheet
AdvanceBio β-N-Acetylhexosaminidase, 5x Reaction Buffer A
Material Safety Data Sheet
AdvanceBio β-N-Acetylhexosaminidase, 5x Reaction Buffer A
Material Safety Data Sheet
AdvanceBio β-N-Acetylhexosaminidase, 5x Reaction Buffer A
Material Safety Data Sheet
