Employee Response
Are the compounds in hand sanitizer what they need to be?
Agilent has a method to ensure they are.
We are all familiar with the advice: wash your hands often with soap and water. When that's not feasible, use hand sanitizer to help prevent disease transmission. While soap and water are effective, they can be less convenient for maintaining germ-free hands in many situations. So hand sanitizer has become an essential weapon for healthcare workers and first responders, as well those working in grocery stores, restaurants, and facilities providing other vital services.
The FDA has released guidance that temporarily allows alcohol manufacturers to produce hand sanitizer products during this Public Health Emergency. In addition to alcohol, these hand sanitizer products require glycerin, hydrogen peroxide, and sterile water.
Distilleries are now stepping in to help combat the hand sanitizer shortage by using the alcohol in their facilities to create their own alcohol-based solutions. Some are packaging it in small bottles while others are encouraging people to bring in their own containers for refills.

Our Employees Respond
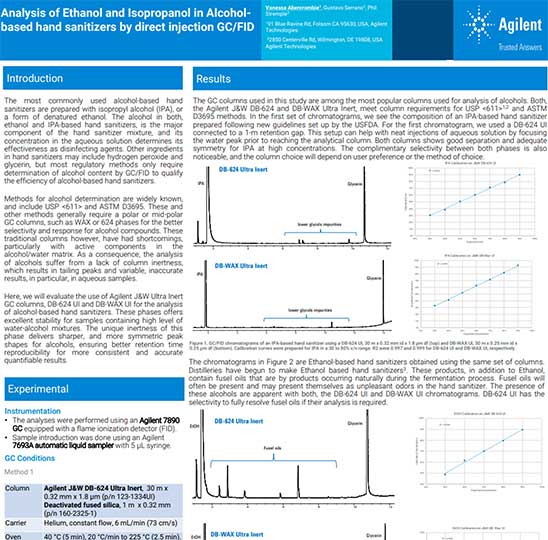
With the increased focus on hand sanitizer manufacture, Agilent has received numerous inquiries on how to properly analyze the component materials, especially methanol contamination. In one response, Agilent employees Vanessa Abercrombie, Gustavo Serrano, and Phil Stremple sprang into action to develop a gas chromatography method for this purpose. Agilent employee Jie Zhang expanded this work to include methanol as a contaminant following USP methodology.
Compound analysis method
Agilent's first method, primarily for determining ethanol or isopropanol content, uses our 7890 GC equipped with a flame ionization detector and our J&W DB-WAX UI and DB-624 UI columns to quantify the alcohols in alcohol-based hand sanitizers. The expanded method uses the 8860 GC equipped with flame ionization detector along with internal standard calibration to meet US Pharmacopiea <611> method recommendations. In both methods, retention times and peak shapes for the alcohols were consistent over the course of several direct sample injections, even in the presence of water being injected as a solvent.
These column phases offer excellent stability for samples containing high levels of water–alcohol mixtures. The unique inertness of this phase delivers sharper and more symmetric peak shapes for alcohols, ensuring better retention time reproducibility for more consistent and accurate quantitative results.
Resources
- Temporary Policy for Preparation of Certain Alcohol-Based Hand Sanitizer Products During the Public Health Emergency (COVID-19), U.S. Department of Health and Human Services, Food and Drug Administration, Center for Drug Evaluation and Research, Washington D.C. June 1, 2020.
- Centers for Disease Control and Prevention, https://www.cdc.gov/handwashing/show-me-the-science-hand-sanitizer.html (accessed Jun 15, 2020).
- Guide to Local Production: WHO-recommended Handrub Formulations, World Health Organization, April 2010.
An infrared method for ensuring post-production sanitizer quality

It is critical for production facilities to ensure that hand sanitizers have the correct final concentration of alcohols. It is equally important to evaluate purchased hand sanitizer to check for alcohol losses during transportation, fraudulent dilution, or tampering at the site of use.
Fourier transform infrared spectroscopy (FTIR) analysis can fulfill the post-production need for fast, reliable, and accurate quantification of alcohol in hand sanitizers and differentiate between alcohol types in unknown samples.
FTIR uses infrared light to scan test samples and observe chemical properties. The analysis requires essentially no sample preparation, and color-coded pass/fail results can be obtained in seconds.

Agilent Cary 630
FTIR

Agilent 4500
FTIR
The Agilent Cary 630 FTIR coupled with Agilent Microlab software enables easy in-lab analysis, while the rugged and portable Agilent 4500 FTIR provides a transportable solution based on the same methodology for analysis in the field.