|
|
Chances are you or someone you know has had an MRI, a medical test in which you lie in an enclosed cylinder while a part of your body is scanned. The result is a microscopic image, which helps doctors determine how to treat you.
What you may not know is that MRI, or magnetic resonance imaging, is based on an analytical technique called NMR, or nuclear magnetic resonance.
Notice how MRI, the more familiar version of the technology, doesn’t include the word "nuclear"? Doctors shortened the term because “nuclear” has a tendency to alarm people who are going into that enclosed cylinder to be scanned. Rest assured, however, that neither MRI nor NMR has anything to do with nuclear weapons or radioactivity. Nuclear simply refers to the nucleus of the atom.
Used in research applications, NMR is one of the technologies developed more than 60 years ago by Varian Associates in Silicon Valley, now part of Agilent. Continually refined over the years, NMR applications allow us to obtain physical and chemical information based on the resonant frequencies of atomic nuclei in a given sample.
With NMR, we can observe molecules and substances in just about any form. They can be liquids, crystals or powders. They don't have to be soluble as they do in other kinds of analysis. Most important, we can observe them in a non-destructive fashion. That is, the sample remains intact for further study (which can be important when studying living organisms).
A noteworthy technology
|
 |
|
In 1948, Varian was founded in California’s Silicon Valley with six employees and $22,000 in capital. One of Varian’s first two products was an instrument that measured the gyro-magnetic ratio of certain atoms. This effect later became known as nuclear magnetic resonance.
In 2010, Varian was acquired by Agilent. Its NMR solutions are now part of Agilent’s Research Products Division in the Life Sciences Group.
Over the years, six Nobel Prizes have been awarded to scientists for their work in NMR and MRI:
- 1943 Physics: Otto Stern (USA)
- 1944 Physics: Isidor Rabi (USA)
- 1952 Physics: Felix Bloch and Edward Purcell (USA)
- 1991 Chemistry: Richard Ernst (Switzerland)
- 2002 Chemistry: Kurt Wüthrich (Switzerland)
- 2003 Physiology/Medicine: Paul Lauterbur (USA) and Peter Mansfield (UK)
|
It's elementary!
More than 100 elements have nuclei with magnetically active isotopes. They react measurably when exposed to magnet fields. The energy level of the nucleus will change in proportion to the strength of the magnetic field. With a suitable apparatus, we can observe the transition from one energy level to another.
Some elements have nuclei that are easily observable using NMR. Fortunately for researchers, these elements include hydrogen, carbon, nitrogen, oxygen, fluorine and phosphorous—the building blocks of all life. They constitute 99% of what makes up all humans, animals and plants.
Figure 1: Periodic table of the elements
|
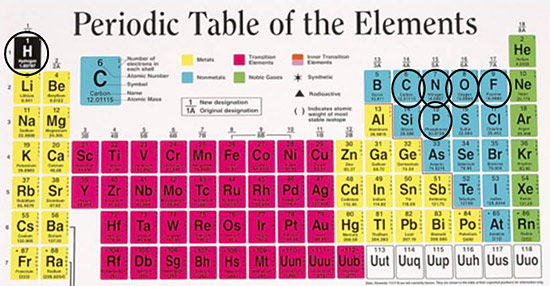
More than 100 nuclei are NMR active. The most commonly measured
include hydrogen, carbon, nitrogen, oxygen, fluorine and phosphorous. |
NMR spectrometry
Here is a simplified schematic of an NMR system. The two yellow cylinders represent a large magnet (some weigh as much as 45 tons) with north and south poles. Researchers insert test samples into the magnet, then create a smaller magnetic field perpendicular to the big magnet. They control the smaller magnetic field via a computer console and record what happens to the sample.
Figure 2: NMR schematic
|

Nuclear magnetic resonance occurs when the nuclei
of certain atoms are immersed in a static magnetic field
and exposed to a second oscillating magnetic field. |
Here is another view of an NMR system. On the right, we have the big magnet (taller than an adult person in some cases). On the far right is a probe containing a tiny sample (about half a milliliter) that is inserted into the magnet. On the left is the NMR console, which creates and controls the second, smaller magnetic field. See more details on How NMR Works.
Figure 3: NMR Spectrometer
|

Researchers use a probe to insert test samples into the center of a superconducting magnet. A computer console generates radio-frequency pulses that excite the sample's nuclear spins away from their normal state of thermal equilibrium. See more details on How NMR Works. |
NMR applications
Unlike many other technologies, NMR can be applied to the study of both chemistry (small molecules) and biology (large molecules). The flexibility of NMR enables us to analyze solids, liquids, liquid crystals and even nano materials. We can even analyze squishy materials such as gels, resins or tissue samples, which are hard to analyze with other techniques. All this with little or no sample preparation—a costly and time-consuming aspect when using other techniques.
In biological NMR, the technology can determine the structure of proteins and nucleic acids in complex molecules for areas like drug discovery. NMR is the only technique that allows us to perform this analysis within a living organism. Consequently, it is also used in academia and pharmaceutical research. Most, if not all, chemistry, bio-chemistry and medical schools use NMR spectrometry.
Most of the proteins in the human body are bound by membranes, which makes them difficult to study, even with NMR. NMR applications in biological solids—currently limited mainly to academia—will likely become widespread as the applications become easier to use.
The full range of NMR applications includes the following:
Pharmaceutical
Drug manufacturers use NMR spectroscopy to measure the composition of their products and verify that they’re making the correct compounds. Sometimes the act of compressing a drug into pill form changes the stability or therapeutic effect. The only way to determine this is by analyzing the pill using NMR. NMR is well-suited to continuously analyze process streams in real-time.
Energy
The continuous, real-time feedback provided by NMR makes it useful for monitoring process streams in hydrocarbon refineries and other fuel-production facilities.
Polymer science
Technicians use NMR spectroscopy to analyze the production of polymers such as plastics and lubricants, where NMR’s wide dynamic range and ability to function over a broad temperature range is advantageous.
Structural biology
Academic labs use NMR spectroscopy to analyze large molecules such as proteins and nucleic acids for disease research and drug discovery. NMR and X-ray diffraction are the main tools for determining the structure of biomolecules and how these structures interact. For example, researchers can determine the 3-D structure of an enzyme and then expose it to a library of chemical compounds to look for changes.
Structural genomics
NMR, in combination with next-generation sequencing, is the mainstream method for determining the 3-D structures of proteins encoded by particular genomes. Because a protein’s function is largely determined by how it is folded, this type of experiment is frequently performed to identify potential drug targets.
Metabolomics
This is the study of metabolites, the chemicals produced by the cells of every plant and animal. Researchers study the metabolome—a snapshot of all the metabolites in a given bio-sample, such as blood or urine —to understand the mechanisms of disease and health, and to find new therapeutics. NMR can identify all metabolites simultaneously and study them noninvasively. With informatics software from Agilent, researchers can even combine data from other disciplines such as genomics and proteomics to gain a holistic understanding of biological systems.
Food science
NMR’s ability to analyze mixtures helps determine the composition and condition of foods and their ingredients. Is a shipment of canola oil too old to use? Is high-volume pet food production within specs? NMR spectroscopy can answer these and other food-related questions on a continuous real-time basis.
Forensics
NMR can determine the origins of illicit drugs or counterfeit pharmaceuticals based on their composition. This information can help identify distribution channels and who handled the materials. Because the procedure is straightforward, a technician with average expertise can consistently produce evidence that meets courtroom requirements.
Environmental
Did an oil slick originate from an undersea production platform or from a ship, and if so, which one? Because NMR is a highly quantitative technique, investigators use it to answer many environmental questions.
Material science
When paint dries, do the solids change? Scientists use NMR to evaluate many types of solid chemicals, catalysts, films, coatings and nanomaterials. Researchers use it to developf new materials, and manufacturers use it to control quality.
Synthetic chemistry
Combined with mass spectrometry, NMR is indispensable in the synthesis of molecules. It’s how scientists know exactly what they’ve made; and how they determine whether a reaction is completed or not.
Clinical/ Diagnostics
Clinics use NMR to conduct a number of diagnostic tests. For example, they use NMR to identify metabolic markers for diabetes and cardiovascular disease.
Midrange NMR markets
Midrange NMR is defined as 400–600 MHz, covering myriad liquids applications in the academic, pharmaceutical, food and chemical industries. Often, midrange systems can be found in shared facilities where they serve a range of applications.
Academic and government
Most NMR users work in academic institutions or government research labs. Applications cover the entire range described above, and span from teaching to method development.
Pharmaceutical industry
NMR is frequently found in pharma workflows, particularly in drug discovery, drug development and ADME, as well as manufacturing.
Food industry
NMR is a powerful tool to address food quality, origin and purity due to its strength in quantitation and identification of samples, not to mention minimal sample preparation. NMR is uniquely capable of quantitation without standards covering a vast range of samples.
Chemical industry
NMR is instrumental in analyzing and manufacturing chemicals and materials such as polymers. Applications range from confirmation of purity to confirmation of structures.
November 2013
|




