Agilent 101: An Introduction to Optical Spectroscopy
|
|

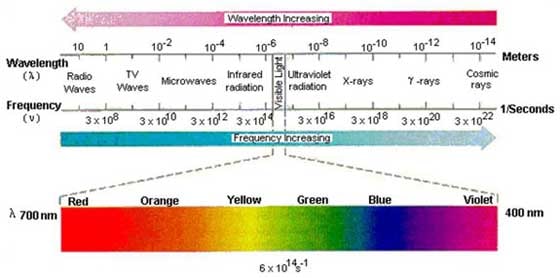
The term "spectroscopy" was first coined in 1666, when Sir Isaac Newton demonstrated the dispersal of white light through a prism into different colors. Contemporaries thought they were looking at ghosts and called themselves "ghost watchers." "Ghost" in Latin is spectrum, while "watcher" in Greek" is scopos – hence the term "spectroscopy." Spectroscopy is basically the measurement of the interaction of light with various materials. By analyzing the amount of light absorbed or emitted by a sample, we can determine what it's made of and how much of it there is. We tend to think about light as being visible, but it's much more than that. The spectrum of electromagnetic radiation ranges from long-wavelength radio waves, to microwaves and infrared light, to visible light (a very small part of the electromagnetic spectrum), to ultraviolet light, to higher energy X-rays, gamma rays and cosmic rays.
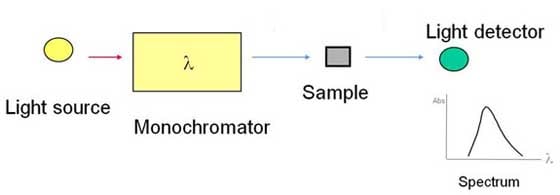
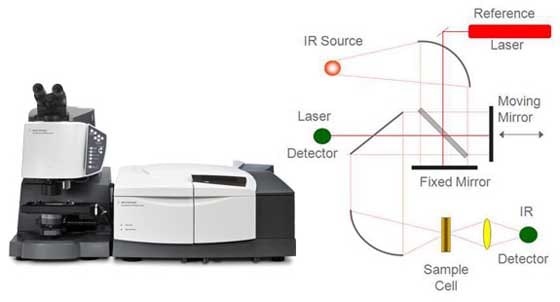
The areas of light used by Agilent's optical spectroscopy instruments range primarily from infrared through ultraviolet. Basically, two things can happen when light hits a sample. We either have absorption or we have emission. In absorption, the sample absorbs some of the energy from the light, which is what happens when your sunglasses stop the ultraviolet rays in sunlight from getting to your eyes. Emission occurs when we hit a sample with some light and it emits light of a different wavelength. This explains phenomena such as fluorescence, luminescence and phosphorescence. Emission is what happens when you wear a white shirt that has been washed in a detergent containing chemical brighteners. It absorbs ambient light and gives off a slight blue hue to make it look cleaner. A clever idea from detergent manufacturers. The effect of light on a sample depends on the wavelength of the light, the intensity of the light, and what it's doing to the molecules or atoms of the sample. Here, in a very simple form, is a schematic of a spectrometer. First, we have a light source. Second, we have some sort of device that can select a specific wavelength for that light. It can be a monochromator, a polychromator, an interferometer, or even something as simple as a filter. Third, we have a means of presenting our sample to the spectrometer.
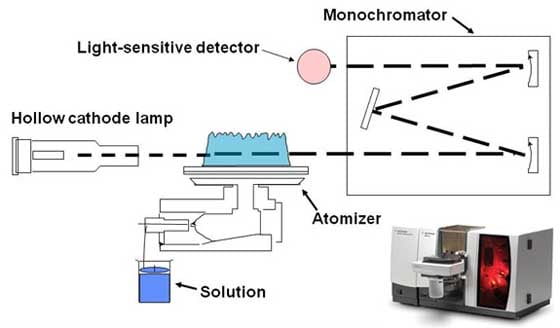
We direct the light onto the sample. We know the intensity and wavelength of that light. Then we use a detector of some sort to measure what comes out the other side. What we will see is a spectrum. We see a change in the absorbance or intensity of that light as we move across the spectrum and change the wavelength. Based on these fundamentals, we have a range of different spectroscopic techniques available to us as scientists and engineers. Atomic spectroscopyThe first of these is atomic absorption spectroscopy. AA was first brought to prominence as an analytical technique by the CSIRO [Commonwealth Scientific and Industrial Research Organization] and Techtron in Australia. Techtron was acquired by Varian, which in turn was acquired by Agilent; that's how the Melbourne part of our company began back in the early 1960s. Atomic absorption is based on the fact that an atom in the vapor state will absorb light of certain frequencies as a unique characteristic of that specific atom. We start with a hollow cathode lamp, which is basically a glass lamp with a metal cathode in it. We effectively heat it and it emits lines of a certain wavelength. These wavelengths are characteristic of the cathode material. If it's copper, for instance, it will emit lines at a number of different wavelengths characteristic of copper. Then we take our sample, which is generally in a liquid solution, and we nebulize it. We suck it up into a flame of a couple thousand degrees, which atomizes the sample. Now we have gaseous atoms that we pass through the light path of the spectrometer. If those atoms are copper, they'll absorb those wavelength lines. As we aspirate the sample, we'll see a decreasing intensity because the atoms are absorbing light. We can relate that decrease in intensity to concentration. And that's how we measure what is in the sample.
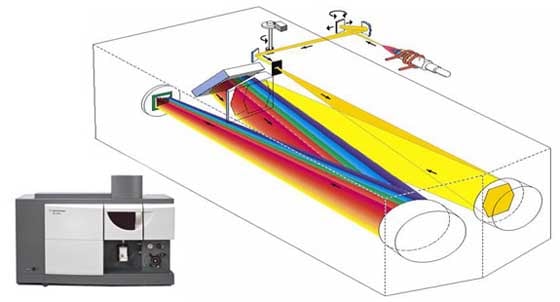
Atomic absorption is a very robust technique. It's used a lot in the mining and mineral industry. If you want to know the concentration of an element – it may be copper, zinc or calcium for instance – then atomic absorption is a good way to do it. Another analysis technique is inductively coupled plasma optical emission spectroscopy. ICP-OES generally uses an argon plasma to excite the atoms in solution. Again, we take a sample that's in a liquid form and we nebulize it. We suck it up and spray it into a plasma, which is about twice the temperature of the flames used in atomic absorption.
In this case, we're talking about emission, not absorption. The sample goes into the plasma, which is very hot. It absorbs that energy and emits light. Again, based on the nature of the sample, we can predict the wavelengths it will emit. We can set up our polychromator to look for those wavelengths and measure how much of the material we're looking for is there. We can relate the intensity of the wavelengths to the concentration in the sample. The advantages of ICP over AA are that we can measure more elements and we can measure them all at once. Atomic absorption is a sequential technique; we have to measure one element after the other by changing the lamp. With ICP, as long as we've got the wavelength range in our polychromator and our detector, we can measure a whole range of substances at once. Molecular spectroscopyNow let's move from the elemental or atomic techniques – AA and ICP – into what we call the molecular techniques. UV-Vis spectroscopy covers the ultraviolet-visible range of the spectrum. By adding another grating and another detector to the instrument, we can extend the wavelength range into the near-infrared with UV-Vis-NIR spectroscopy. Agilent's Cary line of UV-Vis-NIR spectrometers dates back to Howard Cary, the creator of the first commercial recording UV-Vis spectrophotometer in 1948.
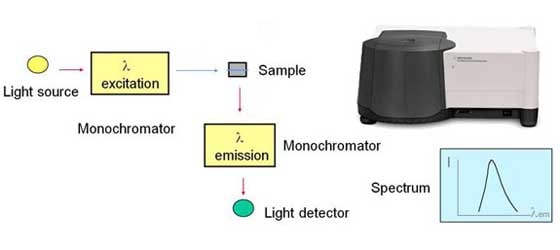
Another technique is fluorescence spectroscopy. Some molecules can take high-energy ultraviolet light, absorb it, and then emit it back out at lower-energy, longer wavelengths. This is the luminescence we see in jellyfish. We also see it in the glow-in-the-dark sticks that people use in nightclubs or when camping.
Finally, there is Raman spectroscopy. This is a complementary technique to infrared spectroscopy. Both technologies examine changes in vibration and rotation at the molecular level. But while infrared measures the amount of IR light absorbed, Raman measures the amount of light scattered. The two techniques are complementary in that they can tell you different things about a molecule.
Spectroscopy applicationsWhere are optical spectroscopy products used? Key markets include industrial, chemical, petrochemical, environmental, food and agriculture, metals and mining. Take the case of a mining company. They want to know how much gold is in their ore. They hope it's the major constituent. But they may also be looking for other mineral components that affect their refining process, that make it less efficient. Food and beverage customers are looking for essential elements in their products – things such as calcium, magnesium and toxic heavy metals in foods, or cadmium and mercury in shellfish. The U.S. Air Force and other customers running big fleets of heavy machinery use our instruments to look at metals in their oil. This helps them determine when they need to change the oil in an engine. On the materials side of things, companies such as Samsung use our equipment in the production of flat-panel displays. A couple of years ago, there was a scare at Mattel in China involving the amount of lead in the paints they were using on their toys. As a result, we sold a lot of ICPs into Mattel's supply chain. In biomedical and pharmaceutical markets, there's a lot of interest at the moment in diseases, pathogen identification and biological screening – to identify cancerous cells, for instance. There's a big push all over the world to use infrared to do that. What does the future hold? Everywhere we go, we hear that smaller is better. Our customers want to take the analyzer to the sample. They want answers fast and don't want to have to set up a lab. It's expensive, it takes space and they've got to maintain it. This was one of the major factors in our recent acquisition of A2 Technologies' assets, where Agilent gained patented technology and portable FT-IR solutions. In the future, our customers will demand distributed detector networks with distributed results. Looking ahead, there are lots of opportunities for synergy between the Melbourne organization and the Chemical Analysis and Life Sciences groups. There is a huge value in Agilent's central research laboratory. We are also collaborating with the Electronic Measurement Group in areas as diverse as high-precision optics manufacture and microwave spectroscopy. As the former Varian and A2 teams get to know the Agilent organization, we get increasingly excited about possibilities for future innovations in spectroscopy.
April 2011
|