Access Agilent eNewsletter, January 2015
>> Update My Profile | Subscribe to Access Agilent | Article Directory

Agilent solutions to optimize workplace drug testing
By Joan Stevens
Agilent Sample Prep Applications Scientist
Many workplace-screening tests for drugs of abuse are done by mass spectrometry (MS) with gas chromatography (GC) or liquid chromatography (LC). At times, you may need to update existing standard operating procedures to achieve lower detection limits, improved linearity in the lower or upper concentration range, calibration range extension, limited sample volume, or to correct frequent replacement of the analytical column. In this article we offer some examples for optimizing forensics sample prep method development.
Better sample prep method development with Agilent Bond Elut Certify
The first step in solid phase extraction (SPE) method development is to understand the characteristics of the target compound. We looked at phencyclidine (PCP), a relatively basic and hydrophobic drug. Ideally, using cation-exchange and hydrophobic interaction would be the best choice for SPE of this drug. Agilent Bond Elut Certify mixed-mode solid phase extraction has a good balance of both hydrophobic and cation-exchange characteristics and is well-suited for PCP analysis. Agilent Bond Elut Certify exhibits a variety of sorbent-analyte interactions so it can be used both for general clean-up and specific extractions for instrumental confirmation of drugs and metabolites.
 Enlarge
Enlarge
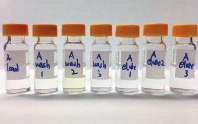
Figure 1. Collection eluates from every step in an SPE method for PCP (from left to right, eluate collections of sample loading, wash 1, wash 2, wash 3, eluate 1, eluate 2, and eluate 3).
 Enlarge
Enlarge
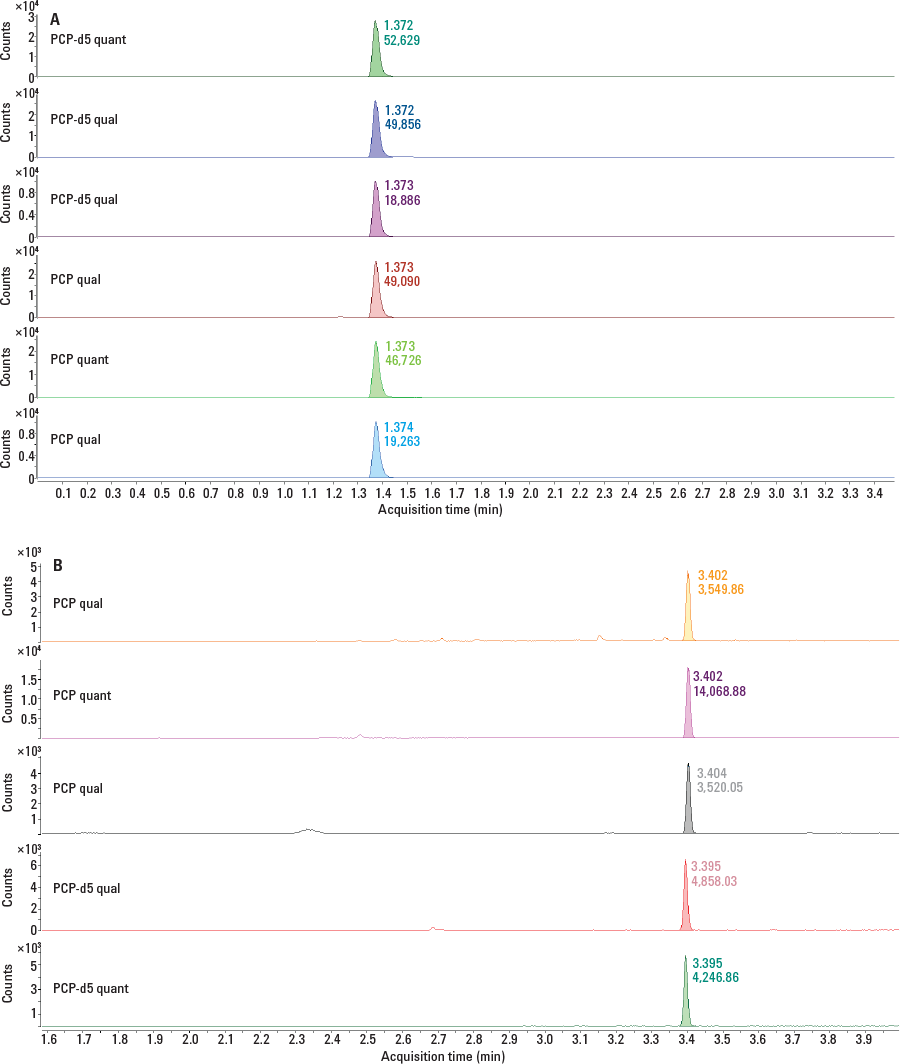
Figure 2. Chromatograms of phencyclidine in urine at the cutoff level (25 ng/mL) by LC/MS/MS (A) and GC/MS with pulsed splitless injection (B).
 Enlarge
Enlarge
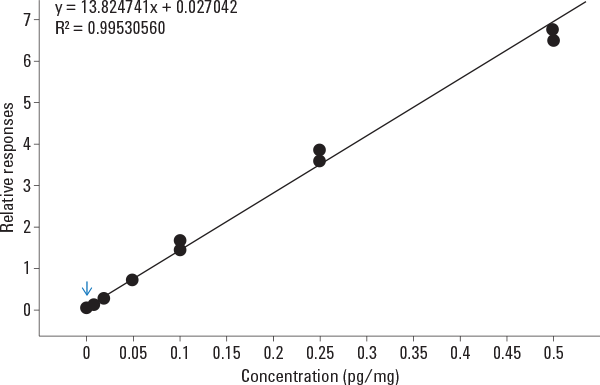
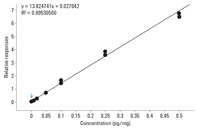
Figure 3. Calibration curve for THCA spiked into hair samples at 0.002, 0.01, 0.02, 0.05, 0.1, and 0.5 pg/mg of hair.
The conventional SPE workflow consists of conditioning, equilibration, sample loading, washing, elution, evaporation, and reconstitution. The most effective optimization is achieved in the washing and elution steps. During sample loading, compounds of interest bind to the sorbent material, along with interferences. The purpose of the washing step is to remove these interferences. The elution step then recovers the target compound by interrupting the interaction between it and the SPE sorbent. Maximum removal of interference, while maintaining optimal recovery of the target compound, is key to successful method development in SPE. To verify method optimization, every step from sample loading to elution needs to be collected and analyzed by chromatography. Figure 1 shows the sample vials at different stages of the washing and elution process in an investigation of PCP in urine.
The wash 3 vial was visually clear and the chromatogram from wash 3 did not have the target compound peak; hence, wash 3 can be skipped. Eluate 3 did not have a significant amount of PCP, which indicated that eluate 2 and 3 could be combined into a single elution step to save on solvent use and time.
Method optimization with Agilent solutions
We then took advantage of the optimized SPE method to analyze PCP by SAMHSA (U.S. Substance Abuse and Mental Health Services Administration) guidelines. We used LC/MS/MS with an Agilent Pursuit XRs Ultra Diphenyl column in our Agilent 1260 Infinity LC with the Agilent 6460 Triple Quadrupole LC/ MS. We then compared the results, using the same samples, with GC/MS by split or splitless injection using an Agilent J&W DB-5ms Ultra Inert column in an Agilent 7890B GC with the Agilent 5975T LTM GC/MSD. The results in Figure 2 demonstrate that performance was independent of the instruments and columns used. You can read the Agilent Application Notes 5991-4695EN and 5991-4575EN for more details on these method improvements.
Detecting THC in hair using Agilent 7000B Triple Quadrupole GC/MS
Testing hair has been practiced for over 50 years. Because many drugs are well preserved in hair, it is possible to detect drug use over longer periods compared with other biological matrixes. Workplace programs often include hair testing due to the ease of collection, difficulty of adulteration, and longer detection times.
Marijuana is one of the drugs tested most often in forensic and drug screening applications. The parent compound, tetrahydrocannabinol (THC), is found in higher concentration in hair samples, but detection of the acid metabolite 11-nor-D9-tetrahydrocannabinol-9-carboxylic acid (THCA) is preferred to eliminate the possibility of potential environmental contamination from marijuana smoke.
In this example, we used an Agilent 7890B GC coupled to an Agilent 7000B Triple Quadrupole GC/MS to provide rapid and sensitive detection of a THC metabolite in hair using 2-D GC. The method took advantage of the lower chemical background and higher sensitivity provided by triple quadrupole MS/MS analysis. Backflush was used to increase robustness, and low thermal mass column modules (DB-1ms LTM and DB-17ms LTM) sped the chromatography process, enabling a run time of 7 minutes and a cycle time of 9 minutes. Multiple reaction monitoring MS/MS analysis delivered excellent sensitivity, with an LOD of 0.002 pg/mg and an LOQ of 0.01 pg/mg. The accuracy of quantification was also good, with an R2 of 0.995 from 0.002 to 0.5 pg/mg of hair (Figure 3). Read the Agilent Application Note 5990-7535EN for more details.
Agilent provides complimentary resources and sample prep solutions for drug testing
The new Essential GC/MS Designer Drug Kit has even more to offer for the rapid and accurate analysis of drugs of abuse, with expanded Agilent ChemStation and MassHunter libraries that include more designer stimulants (‘bath salts’) and synthetic cannabinoids, as well as a broad range of other designer drug classes. If you work mainly with LC/MS, take a look at the new Bioanalysis/Drugs of Abuse Quick Reference Method Guide, and for an in-depth treatment of sample prep methods, check out the new Bond Elut Certify and Certify II Methods Manual. Agilent has a range of sample prep solutions to support your analytical needs, so be sure to check our sample prep pages.
For Forensic Use.
>> Update My Profile | Subscribe to Access Agilent | Article Directory
Figure 1.

Collection eluates from every step in an SPE method for PCP (from left to right, eluate collections of sample loading, wash 1, wash 2, wash 3, eluate 1, eluate 2, and eluate 3).